How to perform ELASTA-Clip: lacerating M-TEER devices to enable TMVR
Recurrent mitral regurgitation after M-TEER can create a challenging scenario. Discover the ELASTA-Clip technique and its key steps to enable TMVR!
Frequency of the problem:
Expert level:
Summary
Mitral transcatheter edge-to-edge repair (M-TEER) represents an established and widely adopted treatment approach for patients with symptomatic severe mitral regurgitation (MR), who are at increased surgical risk1.
Even though procedural techniques are refined and success rates are high, relevant MR may recur despite sufficient insertion of both mitral valve leaflets.
Repeat M-TEER procedures are technically challenging and often anatomically unfeasible. Electrosurgical LAceration and STAbilization of Clip / M-TEER devices (ELASTA-Clip) has been introduced as a novel innovative technique to intentionally detach an M-TEER device from the anterior mitral valve leaflet to enable subsequent transcatheter mitral valve replacement (TMVR)2.
This Toolkit article aims to provide you with a reproducible step-by-step guide of the ELASTA-Clip procedure, explaining the technical concept of the procedure, providing a list of necessary material, and highlighting key procedural steps.
The problem
Even with ever increasing experience of M-TEER operators and continued device iterations, following successful initial M-TEER relevant MR can recur, then creating a challenging interventional scenario.
Potential repeated M-TEER procedures are technically demanding, as there is only little maneuverability left and the prior devices in place may hamper proper imaging.
Furthermore, given small valve areas and already heightened gradients, such procedures are often unfeasible to be performed.
TMVR as an alternative transcatheter MR treatment is more and more becoming available, however, M-TEER devices in place with the created double-orifice anatomy render implantation of a TMVR prosthesis impossible.
Principal idea
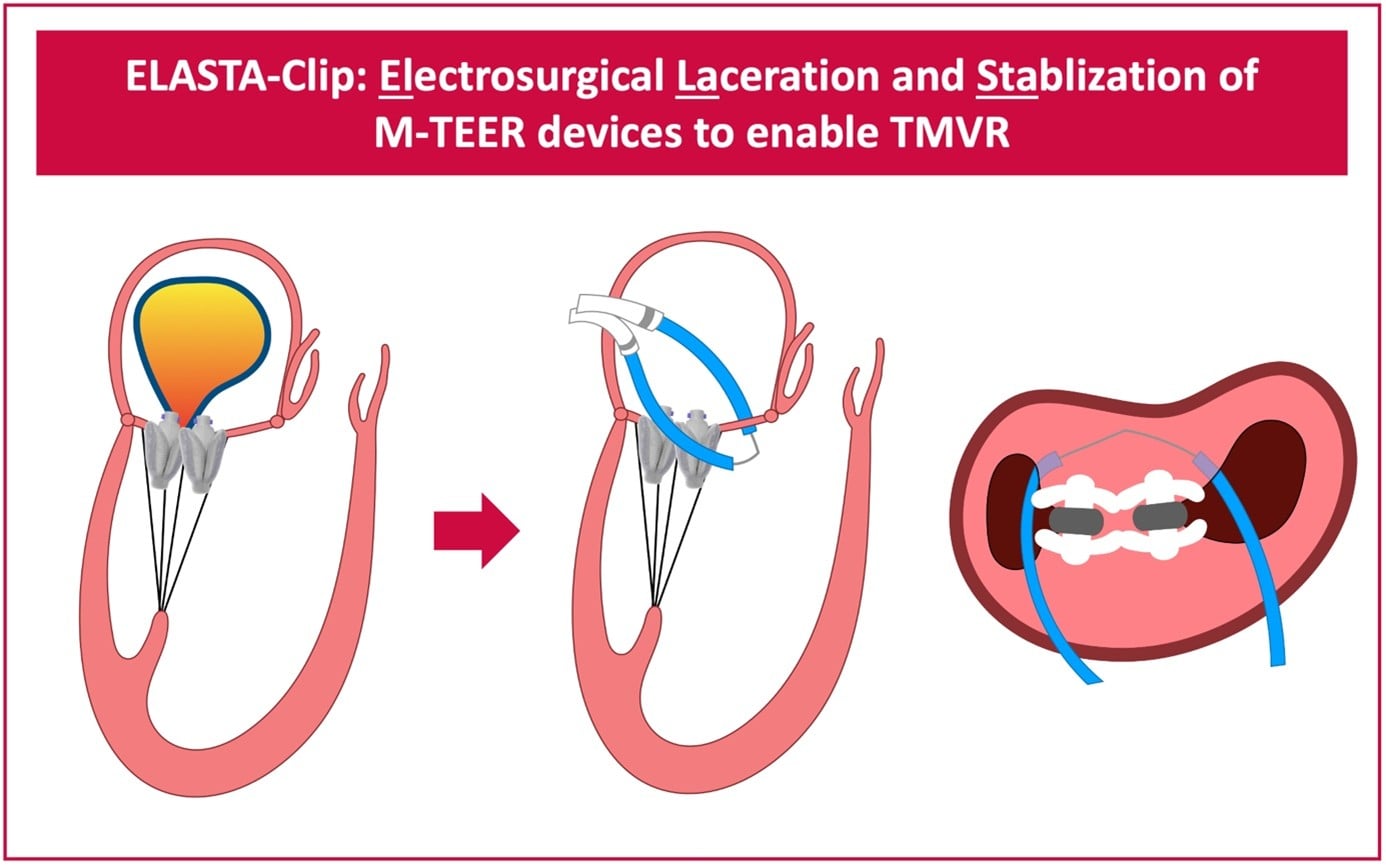
The main idea of the ELASTA-Clip procedure is to burn the created M-TEER tissue bridge by means of electrosurgery to recreate a single-orifice mitral valve and give space for subsequent TMVR.
By using an electrified wire that is looped around the M-TEER devices anteriorly, the anterior mitral valve leaflet is cut and a single leaflet device attachment scenario, with the M-TEER device remaining attached to solely the posterior mitral valve leaflet, is created.
This clears the path to then implant a TMVR prosthesis, which will push the M-TEER devices towards a posterior portion of the left ventricle.
Figure 1: Schematic of the ELASTA-Clip procedure (here with two PASCAL M-TEER devices in place as in the exemplary case presented below)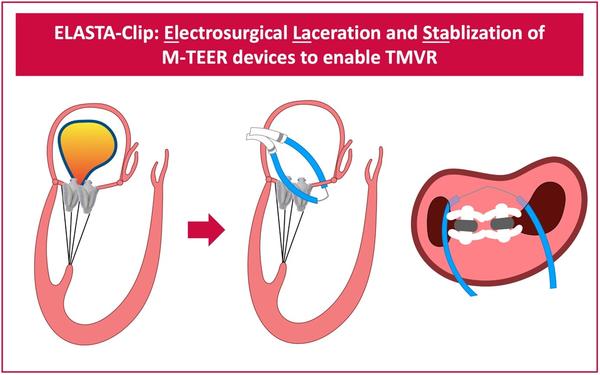
Material needed
- Large-bore inflatable-hub introducer sheath options, 24-26 Fr
- DrySeal Flex sheath
- Allows for multiple parallel guiding sheaths through a single access site
- Transseptal puncture options
- Mullins or Swartz SL transseptal sheaths / Brockenbrough curved needle
- Abbott transseptal kit
- NRG transseptal kit
- TSP Crosser
- VersaCross
- Steerable introducer options
- Agilis NxT (8.5 Fr / Medium Curl)
- Nagare bidirectional steerable sheath (8.8 Fr)
- Oscor Destino Twist (8.5 Fr)
- Guidewires
- ESSENTIAL: Astato XS 20, 300 cm x 0.014’’ wire
- 0.018’’ standard and exchange length wires (e.g., V-18 ControlWire)
- 0.025’’ / 0.035’’ standard and exchange length wires
- Hydrophilic coated soft guidewires
- Guide catheters
- Coronary guide catheters (JR4, IM, MP)
- Snare options
- Merit EN Snare Endovascular Snare System 18-30, 27-45
- Atrieve Vascular Snare Kit (multi-loop) 18-30 mm
- Amplatz Goose Neck Snare Kit 30 mm
- Other useful material
- Balloon tipped wedge catheters, end-hole, 0.035’’ / 0.025’’, 110-cm length
- PiggyBack wire converter, coronary micro-guide catheter like Terumo Finecross
- ESSENTIAL: electrosurgical generator and cautery pencil
- D5W (5 % dextrose in water) IV fluid
- Intra-aortic balloon pump as back-up
Step-by-step method
Step 1: access and transseptal puncture
- One arterial and one venous femoral access sites:
- venous access for transseptal crossing and then ELASTA-Clip procedure with both steerable sheaths via large-bore sheath;
- one arterial access for pigtail to measure LV pressures.
- Alternatively, use 2 venous accesses.
- An additional arterial access can be considered as back-up if circulatory support would be needed.
- Transseptal puncture, aiming at slightly anterior position, give enough working range above the mitral valve but doesn’t aim to high with a very posterior position.
Step 2: transseptal positioning of steerable sheaths
- After transseptal puncture, use sheath and additional wire to place subsequent devices through the same transseptal access.
- Two steerable sheaths are brought transseptally and are placed above the two orifices of the double-orifice structure of the mitral valve.
- To precisely position the sheaths they can be angulated, rotated to steer them anterior or posterior and can be placed at a more medial or lateral position.
Step 3: traverse of mitral valve orifices
- Through the Agilis sheaths, using Swan-Ganz balloon-catheters to avoid chordal entrapment: first, the medial, and then, the lateral orifice of the double-orifice mitral valve are traversed.
- After successful traversal, guidewires are used for catheter exchange and a 6 Fr JR4 guide catheter in the lateral and a 6 Fr JR4 guide (6 Fr multipurpose is also possible, depending on anatomy) catheter in the medial orifice.
Step 4: wire snaring to create loop
- The Astato 0.014’’ coronary wire is inserted into the left ventricle via the medial cath/sheath system; via the lateral JR4 cath/sheath system the Snare system is introduced
- The Astato wire is advanced towards the lateral snare and once the basket of the snare is exposed it is used to capture the wire, creating a loop
Step 5: wire externalisation and creation of “Flying-V”
- The Astato wire that has created a loop around the M-TEER device(s) is externalised; then, using a scalpel, the wire is denuded at the proximal end and the wire is kinked (with the denuded surface at the inner part of the kink) to create the so called “Flying-V”.
- A wire converter or microcatheter is brought onto the wire and positioned close to the Flying-V to further isolate the wire when electrosurgical energy is applied; then, the wire with the Flying-V is brought back into place around the M-TEER device(s).
Step 6: precise positioning of the “Flying-V” at the anterior mitral valve leaflet
- It is essential to ensure that the Astato wire is looped around the M-TEER device(s) at their anterior aspect to then lacerate the anterior mitral valve leaflet (the principal idea is that the device(s) remain attached to the posterior mitral valve leaflet as otherwise at the anterior leaflet they would obstruct the LVOT); here, deflexing and rotating anterior with the steerable sheaths, as well as carefully advancing the flying V more apical are helpful troubleshooting techniques.
- After precise positioning of the wire, before electrosurgical burn, the TMVR system should be prepared, to allow timely and straightforward implantation following leaflet laceration.
Step 7: electrosurgical laceration of the anterior mitral valve leaflet
- The back end of the Astato wire is connected to a cautery pencil
- Using an electrosurgical generator continuous 70 W duty current is applied to the Astato wire and, while the catheters are flushed with D5W (5 % dextrose in water) to hinder alternative current paths other than at the contact of the Flying-V with the anterior mitral valve leaflet, by applying continuous gentle pull forces to the two catheters the anterior mitral valve leaflet is burned; make sure to use Torquers and lock tight to fixate Astato Wire and guides.
-> After electrosurgical leaflet laceration, TMVR is performed as per standard fashion of the respective device; then, pushing the M-TEER device attached to only the posterior mitral valve leaflet to rest towards a posterior pocket of the left ventricle.
Points of specific attention
- Laceration of MitraClip as well as PASCAL devices is possible, laceration of up to three M-TEER devices has been reported, however, central position of the tissue bridge is essential3,4
- Wire position at the anterior mitral valve leaflet is key; combined check via fluoroscopy and TEE is mandatory
- For an effective electrosurgical burn isolation of the wire (using a wire extension), precise focal denudation, insulation with catheters close to the kink of the flying V, dextrose flush during current application, and sufficient loop tension are essential
- An intra-aortic balloon-pump should be ready to support in the case of hemodynamic deterioration after laceration
A word from the reviewer
Commentary
The ELASTA-Clip technique comes at an important moment for transcatheter mitral therapies. As TMVR becomes more widely available, clinicians are increasingly seeing patients with recurrent mitral regurgitation after previous M-TEER. In some of these patients, repeat clipping or surgery is not possible. However, the double-orifice anatomy created by the clips may also prevent straightforward TMVR. Curio and Adam provide a practical solution for this situation.
Where ELASTA-Clip fits in the treatment pathway?
Current guidelines recognise M-TEER as an important treatment for severe MR in high-risk patients, but they do not yet clearly address what to do when M-TEER fails and TMVR is considered. ELASTA-Clip helps fill this gap. The technique uses electrosurgery to cut the tissue bridge created by the clips and recreate a single mitral orifice, allowing TMVR to be performed afterwards. Early multicentre experience suggests that ELASTA-Clip followed by TMVR can be performed successfully in carefully selected patients with failed M-TEER. Importantly, ELASTA-Clip should not be seen as a standalone therapy. It is best considered a preparatory step before TMVR, and both procedures should be planned together.
Importance of CT planning
Careful CT planning is essential. CT imaging helps assess the thickness and position of the tissue bridge, plan the path of the guidewire across the two orifices, and estimate the risk of LVOT obstruction after TMVR.
Practical points for teams adopting the technique
The most critical moment is the short period between leaflet laceration and valve implantation. During this phase, severe mitral regurgitation may occur and haemodynamics can deteriorate quickly.
For this reason:
- The TMVR system should be fully prepared before starting electrosurgical laceration.
- Continuous haemodynamic monitoring is important.
- Mechanical circulatory support should be readily available if instability develops.
Expanding concept beyond the mitral valve
The concept of clip laceration to enable valve replacement is now extending beyond the mitral position. A tricuspid version of the technique has recently been described, highlighting how electrosurgical laceration of prior tricuspid TEER devices may facilitate subsequent transcatheter tricuspid valve replacement.
Future directions
As TMVR platforms become more widely used, the need for strategies to manage prior M-TEER anatomy will increase. Prospective registries and longer-term follow-up will be needed to better define the safety and durability of the ELASTA-Clip plus TMVR approach.
This Toolkit article provides a clear technical guide to a procedure that may become increasingly relevant as the number of patients with prior M-TEER continues to grow.
References
- Curio J, Kuhn EW, Körber MI, et al. Electrosurgical laceration and stabilisation of three clip devices (ELASTA-Clip) to enable transcatheter mitral valve implantation. EuroIntervention. 2023;19:744-745.
- Khan JM, Gill HS. Burning the Bridge: Tricuspid ELASTA-Clip. JACC Cardiovasc Interv. 2025;18(23):2908-2910.
References
- Praz F, Borger MA, Lanz J, Marin-Cuartas M, Abreu A, Adamo M et al. 2025 ESC/EACTS Guidelines for the management of valvular heart disease. Eur Heart J 2025;46:4635-4736.
- Lisko JC, Greenbaum AB, Guyton RA, Kamioka N, Grubb KJ, Gleason PT et al. Electrosurgical Detachment of MitraClips From the Anterior Mitral Leaflet Prior to Transcatheter Mitral Valve Implantation. JACC Cardiovasc Interv 2020;13:2361-2370.
- Nienaber S, Curio J, Kuhn EW, Wienemann H, Adam M. Electrosurgical-laceration and stabilization of two PASCAL devices using artificial intelligence-based procedural planning: a case report. Eur Heart J Case Rep 2025;9:ytaf202.
- Curio J, Kuhn EW, Korber MI, Baldus S, Khan JM, Adam M. Electrosurgical laceration and stabilisation of three clip devices (ELASTA-Clip) to enable transcatheter mitral valve implantation. EuroIntervention 2023;19:744-745.
Disclosures
- Dr Curio has received research grant support from Boston Scientific; has served on an advisory board for Medtronic; and has received travel support from Laralab and Cardiovalve and JenaValve Technology.
- Dr Adam reports consulting fees from Abbott, JenaValve Technology, Edwards Lifesciences, Haemonetics, Medtronic and Meril.
Authors






No comments yet!