444 results
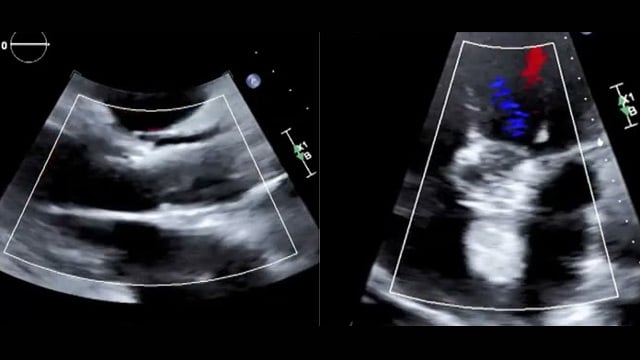
DCB in in-stent restenosis - LIVE Case
07 Apr 2026 – From GulfPCR-GIM 2025
A 60-year-old male with hypertension, type II diabetes, and moderately impaired LV function (45%), has a history of LAD PCI treated with three Cypher stents in 2006. He underwent further treatment for LAD in-stent restenosis in 2012, 2020, and 2022 using a new stent and DCB....
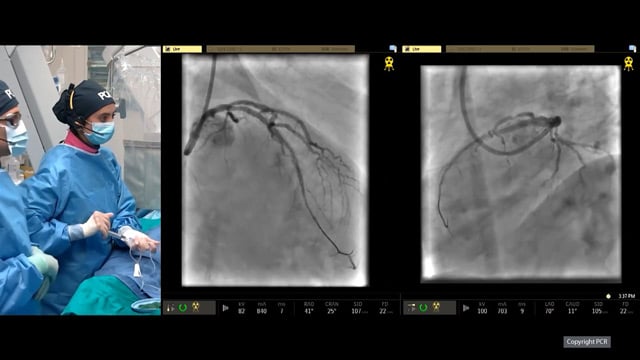
Coronary calcification versus thrombus: a deceptive ambiguity revealed in the aftermath of embolisation
07 Apr 2026
Discriminating between coronary calcification and thrombus in acute settings remains a diagnostic challenge with significant implications for interventional decision-making.

Author

Author

Author
Bioresorbable vascular scaffold versus metallic drug-eluting stent in patients at high risk of restenosis: final 7-year results of the COMPARE-ABSORB trial
10 Mar 2026
This study provides the longest available follow-up for first-generation bioresorbable vascular scaffolds and fails to demonstrate any late clinical advantage after complete scaffold resorption.

Reviewer

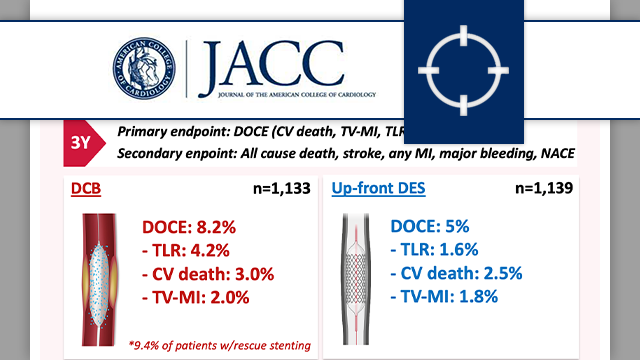
Drug-coated balloon angioplasty vs up-front stenting for de novo CAD: 3-year follow-up of REC-CAGEFREE I trial
10 Mar 2026
The 3-year follow-up of the REC-CAGEFREE I trial demonstrated that a DCB-based strategy with rescue stenting was associated with a significantly higher rate of DOCE compared with contemporary DES implantation in patients with de novo, non-complex coronary artery disease.

Reviewer

Reviewer
Drug-eluting balloon in calcified lesions and complex lesions
28 Feb 2026 – From Euro4C Annual Meeting 2026
This session explores the application of drug-eluting balloons (DCB) in managing calcified and complex coronary lesions. Emphasis is placed on identifying appropriate lesions for a DCB-only strategy, the critical role of vessel preparation prior to drug delivery, and the long-term physiological assessment following treatment, supported by...
Imaging to the rescue
11 Dec 2025 – From GulfPCR-GIM 2025
This session highlighted the pivotal role of advanced intracoronary imaging techniques, particularly OCT, in complex case management such as in-stent restenosis and thrombus evaluation. Through real-world cases, it demonstrated how imaging complements physiology to guide therapeutic decisions, including the use of drug-coated balloons beyond native coronary...

Coronary Pot Pourri - 2
11 Dec 2025 – From GulfPCR-GIM 2025
This session presents a diverse range of complex coronary interventions, highlighting advanced techniques and strategies. Topics include the use of ECMO support for high-risk PCI and TAVI in cardiogenic shock, innovative approaches in left main interventions, challenging stent optimisation, and management of patients with combined severe...

Innovations in stentless coronary intervention: from DCB technologies to bioresorbable scaffolds
11 Dec 2025 – From GulfPCR-GIM 2025
This session delves into the evolving landscape of stentless coronary interventions, focusing on the structural diversity of Drug-Coated Balloons (DCB) and the promising future of bioresorbable scaffolds (BRS). It emphasizes the role of intracoronary imaging, particularly OCT, in optimizing these therapies. Attendees gain a comprehensive understanding...

Abstracts - Part 2
11 Dec 2025 – From GulfPCR-GIM 2025
This session compiles recent clinical abstracts addressing diverse topics in interventional cardiology. Highlights include the HYBRID-CTO approach from a prospective case series, the cardiovascular impact of empagliflozin in type 2 diabetes, off-label use of intravascular lithotripsy for resistant in-stent lesions, and experiences with non-hyperaemic pressure ratios...

Beyond the horizon - Shaping the future of cardiology through meaningful innovation in structural heart and coronary - LIVE Case
11 Dec 2025 – From GulfPCR-GIM 2025
This live case–based session explores how innovation is shaping structural heart and coronary interventions.
A real-time TAVI case in a degenerated aortic valve highlights pre-procedural imaging, sizing strategies, procedural decision-making, and post-procedural care.
The session also reviews ongoing and upcoming Myval THV trials, showing how new technologies impact...