A randomized, placebo-controlled trial of chronic total occlusion PCI in stable angina - ORBITA-CTO trial
Reported from ACC.26
Kalaivani Mahadevan provides an in-depth analysis of the ORBITA-CTO trial: a first-ever randomised, double-blind, placebo-controlled trial evaluating CTO PCI in stable angina patients.
She also interviewed principal investigators Sarosh Khan and John Davies following the LBCT Scientific Session at ACC.26 in New Orleans. Watch interview!
Background and trial objective
Interventions for chronic total occlusion [CTO PCI] currently carry a 2bB recommendation in North America and a 2aB recommendation in Europe based on symptom and quality of life improvement.1-2 Given the findings of the ORBITA Trial, demonstrating the role of a placebo effect following contemporary PCI for chronic coronary syndrome, the ORBITA–CTO Trial was devised specifically to study the efficacy of CTO PCI beyond that of placebo3-4.
Trial methodology
This was a double-blind, placebo–controlled, randomised trial of CTO PCI.5 Figure 1 outlines the study design.
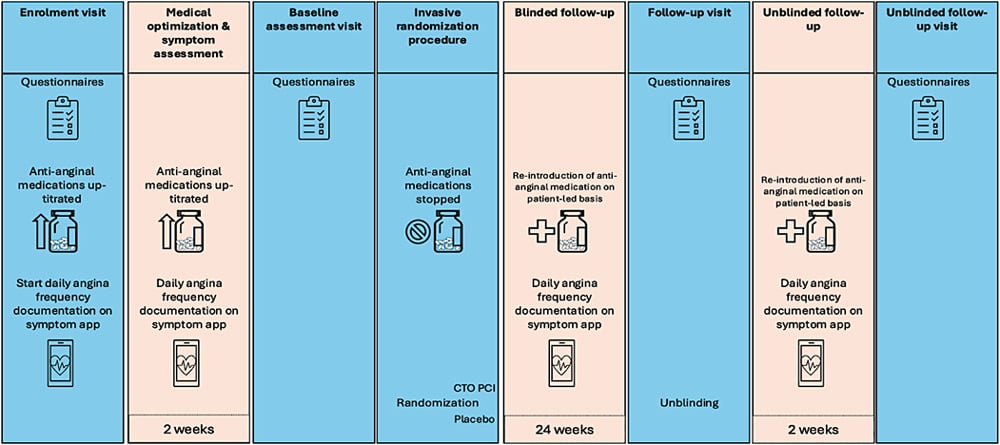
Figure 1 – Study Design (Courtesy of S Khan, ACC slides)
Key inclusion criteria:
- Acceptance for CTO PCI by a specialist operator
- Symptomatic single vessel CTO despite optimal medical therapy [OMT], in a vessel of at least 2.5mm diameter or greater.
- Absence of angiographically ‘significant’ by-stander non-CTO disease [≤50% Left – Mainstem, and/or ≤70% LAD/RCA/Cx/SVG in vessels measuring at least 2mm in diameter]
- Typical angina or exertional breathlessness considered to be angina equivalent
- Clinical evidence of ischaemia and viability in the CTO territory
- J-CTO Score≤3.
Key exclusion criteria:
- Age<18 years
- ACS within prior 4 weeks
- PCI to a non-CTO lesion within 4 weeks
- Clinically significant non-CTO disease requiring revascularisation
- Ischaemia in a non-CTO territory
- Contraindication to drug-eluting stent or dual antiplatelet therapies.
Comprehensive measures were implemented to maintain the integrity of blinding – these are summarised in Table 1 and discussed with the investigators in the Q+A interview.
- Primary efficacy outcome – change in daily angina ordinal clinical outcome scale.
- Secondary efficacy outcomes included: patient-reported change in SAQ physical limitation, angina frequency, angina stability, treatment satisfaction and quality of life [QoL] scores; change in SAQ summary scores; EQ-5D-5L QoL measures and physician-assessed symptom severity via CCS, NYHA and RDS (Rose dyspnoea) scores.
- Pre-specified methodological outcome – fidelity of blinding utilising Bang’s blinding index with a threshold of -0.2 to +0.2.
- In the absence of any prior placebo–controlled CTO trial, sample size was pragmatically derived, with 50 patients providing 80% power and a two-sided alpha of 0.05, allowing for a 10% attrition rate. All analyses were conducted on an intention – to – treat basis.
Table 1: Optimisation of Blinding and Placebo Procedure Measures (produced by K. Mahadevan for PCRonline)
Time masking | All devices reading time [clocks/phones/watches] removed incl. in corridors and Cath lab, digital clocks on screens concealed. Time devices returned 2 hours post-procedure.
|
Patients prepped and recovered in a side room to prevent temporal unblinding by other patients.
| |
Auditory and sensory masking | Sensory deprivation techniques – music via over-the-ear headphones for auditory isolation and deep conscious sedation rendering patient unresponsive to voice or tactile stimulation.
|
Procedural optimisation | Dual access, dual angiography and 60 minutes of simulated PCI in the placebo cohort incl. catheter manipulation C-arm manoeuvres, verbal calls for PCI equipment and inflation and deflation of indeflator.
|
IV adenosine – to measure physiology in the PCI cohort and to generate symptoms in the placebo cohort.
| |
Research Fellow dismissed from Cath Lab pre – randomisation, remaining blinded
| |
Restriction of Interaction | No contact from PCI procedural team after Cath Lab exit with protocolised scripted handover to blinded ward recovery team.
|
Omission of PCI details in patient notes and GP summary | Standardised procedural reports to maintain blinding with secured hand-written reports stored on site for access in emergency only.
|
Standardised discharge summary to GP. |
Results
A total of 50 patients were randomised 1:1 to receive CTO PCI or placebo (simulated PCI). Key patient and procedural demographics are illustrated in Figure 2.
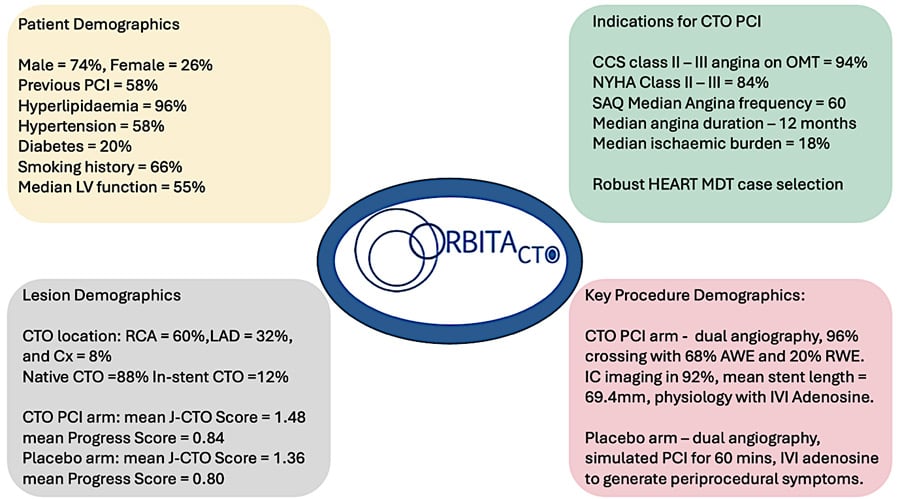
Figure 2: Patient and Procedural Demographics (produced by K Mahadevan for PCR Online)
CTO PCI led to an immediate and sustained improvement in angina symptom score, beyond placebo, [OR 4.38 95% CrI 1.57 – 12.69, Pr(benefit) = 0.996] driven by reduction in angina frequency over a median follow-up of 174 days - Figure 3.
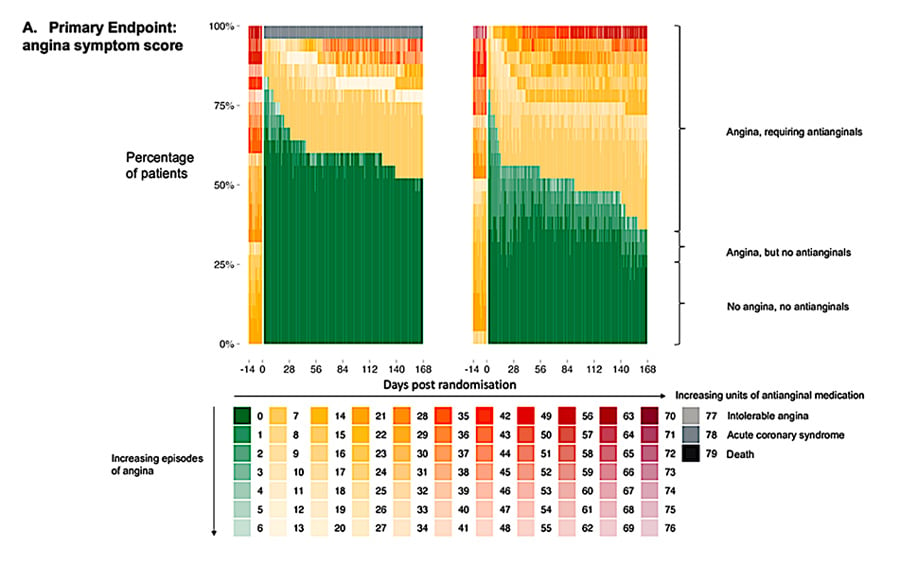
Figure 3: Primary Endpoint – Angina Symptom Score (Courtesy of Khan S et al, JACC reference)
Improvements across SAQ domains including physical limitation [mean difference +13.5 points, Pr(benefit) >0.999] and disease – specific quality of life [mean difference +18.2 points, Pr(benefit) = 0.997] were also observed. Whilst symptom benefit was seen in both cohorts, improvements in SAQ summary score [mean difference +13.7 points, Pr(Benefit) =0.997] and blinded physician-assessed CCS Class [-0.7, 95% CrI -0.27 to -1.13, Pr(benefit)>0.999, Figure 4] were significantly higher in the CTO PCI cohort.
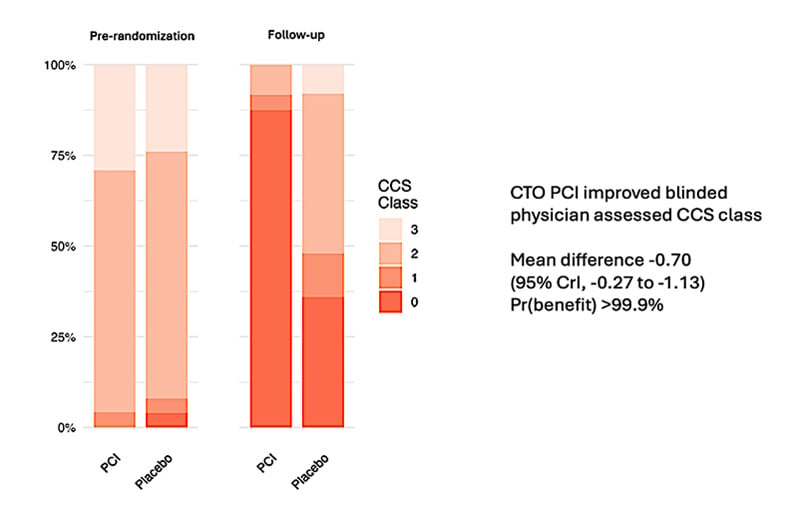
Figure 4: Change in CCS Angina Class, CTO PCI versus Placebo (Courtesy of Khan S et al JACC reference)
Fidelity of blinding was excellent across patient groups (-0.08 in the CTO PCI cohort [95% CI -0.19 to 0.02], and 0.04 in the placebo cohort [95% CI -0.17 to 0.25]), clinical staff and research personnel, Figure 5.
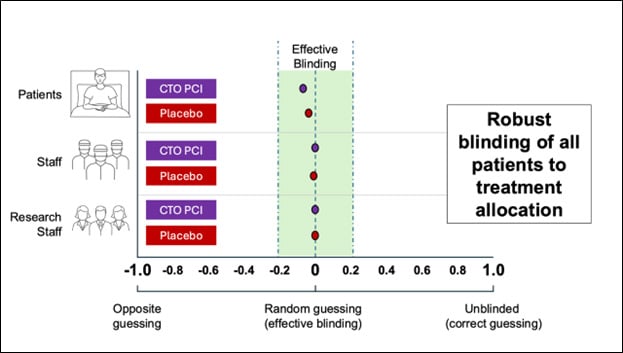
Figure 5: Primary Methodological Outcome – Fidelity of Blinding (Courtesy of Khan S et al. JACC Ref)
Discussion
Prior study of CTO PCI has yielded mixed results regarding the true symptom benefit of CTO revascularisation. Whilst both EURO–CTO (an open label RCT) and OPEN–CTO (a large prospective registry) both demonstrated symptom reduction and health status improvement, the DECISION CTO Trial, conversely, did not find a sustained symptom improvement with CTO PCI.6-8 However, the difference in outcome seen in DECISION–CTO is potentially explained by a high cross-over rate (almost 20% ) the option for PCI in non-CTO lesions, slow recruitment and underpowering.
In addition to extensive blinding, the careful selection of single vessel CTO lesions in the absence of significant by-stander disease, and the exclusion of J-CTO 4-5 lesions to minimise technical failures, which would dilute true treatment effect, has allowed ORBITA-CTO to mitigate the challenges encountered by prior studies. Whilst the external validity of the study is therefore limited to J-CTO 0-3 lesions, these lesions constitute the majority of work encountered by many CTO operators, hence findings will still be generalisable to a large patient cohort in real-world clinical practise. Other limitations include the maturity and procedural volume of the CTO service alongside highly experienced operators which may not be the case in all settings.
Whilst overall patient sample size was modest (n=50), the primary outcome was derived from over 8,600 follow-up days of data. Study strengths included a methodologically exemplary trial design, robust and systematic blinding methods, comprehensive HEART Team MDT for unbiased, consistent decision – making and rigorous and complete follow-up data. Further in-depth discussion and additional points of interest can be found in the Q+A interview.
Conclusion
ORBITA–CTO, the first double-blind placebo-controlled randomised trial in CTO interventions, has demonstrated the immediate and sustained true symptom benefit of CTO PCI, beyond placebo, in a thoughtfully selected cohort of patients. In addition to providing definitive evidence for a long-standing unanswered question in the field of CTO PCI, it has, through its innovative and robust trial design, perhaps set a new methodological benchmark for future study of symptomatic benefit in complex coronary interventions.
Interview with lead investigators John Davies and Sarosh Khan
Acknowledgements
With many thanks to the lead investigators, Dr Sarosh Khan and Dr John Davies from the Essex Cardiothoracic Centre, for their time and participation in our Q+A interview session for this review.
References
- Lawton JS, Tamis-Holland JE, Bangalore S et al. 2021 ACC/AHA/SCAI Guideline for Coronary Artery Revascularization: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. J Am Coll Cardiol. 2022 Jan 18;79(2):e21-e129
- Neumann FJ, Sousa-Uva M, Ahlsson A et al. 2018 ESC/EACTS Guidelines on myocardial revascularization. Eur Heart J. 2019 Jan 7;40(2):87-165
- Al-Lamee R, Thompson D, Dehbi HM et al. Percutaneous coronary intervention in stable angina (ORBITA): a double-blind, randomised controlled trial. Lancet. 2018 Jan 6;391(10115):31-40
- Khan S , Fawaz S, Simpson R et al. The challenges of a randomised placebo-controlled trial of CTO PCI vs placebo with optimal medical therapy: The ORBITA-CTO pilot study design and protocol. Front Cardiovasc Med. 2023 May 3:10:117276
- Khan S, Sajjad U, Fawaz S et al. A Randomized, Placebo-Controlled Trial of Chronic Total Occlusion Percutaneous Coronary Intervention in Stable Angina - ORBITA-CTO
- Werner GS, Martin-Wuste V, Hildick-Smith D et al. A randomized multicentre trial to compare revascularization with optimal medical therapy for the treatment of chronic total coronary occlusions. Eur Heart J. 2018 Jul 7;39(26):2484-2493
- Sapontis J, Salisbury AC, Yeh RW et al. Early Procedural and Health Status Outcomes After Chronic Total Occlusion Angioplasty: A Report From the OPEN-CTO Registry (Outcomes, Patient Health Status, and Efficiency in Chronic Total Occlusion Hybrid Procedures). JACC Cardiovasc Interv. 2017 Aug 14;10(15):1523-1534
- Lee SW, Lee PH, Ahn JM et al. Randomized Trial Evaluating Percutaneous Coronary Intervention for the Treatment of Chronic Total Occlusion: The DECISION-CTO Trial.Circulation. 2019 Apr 2;139(14):1674-1683
Authors






No comments yet!