Redo-surgery versus transcatheter valve-in-valve for mitral bioprosthetic dysfunction: The SURVIV trial
Reported from ACC.26
Yohei Ohno provides his takeaways from the results of the SURVIV trial, which were presented by Dimytri Siqueira during the ACC.26 in New Orleans.
Why this study – the rationale/objective?
Redo mitral valve replacement (rMVR) remains the standard treatment for failed bioprosthetic mitral valves. However, redo surgery carries substantial risk, particularly in patients with prior sternotomy and comorbidities.
Transcatheter mitral valve-in-valve (mVIV) has emerged as a less invasive alternative, but no randomised trial had directly compared mVIV with redo surgery. The SURVIV trial aimed to compare clinical outcomes of mVIV vs. redo surgical mitral valve replacement in patients with failed bioprosthetic mitral valves. It was presented in a Late-Breaking Clinical Trials Session at ACC Congress 2026.
How was it executed?
The investigator-initiated SURVIV trial (Siqueira DA et al. EuroIntervention. 2026 doi: 10.4244) enrolled 150 patients and was randomised in a 1:1 fashion to either mVIV or rMVR. Many participants had underlying rheumatic valve disease. Inclusion criteria were patients with symptomatic severe bioprosthetic valve dysfunction who were eligible for both rMVR or mVIV. This study was conducted in 7 centres in Brazil.
The primary endpoint was a composite of all-cause death or disabling stroke at 1 year. The secondary safety endpoint was a composite of major adverse events within 30 days after the procedure. The mean age of patients was 58 years; mean STS score 3.6%, 72% were women, 56% were NYHA III/IV, 70% had pulmonary hypertension, 50% had atrial fibrillation, and 25% had a history of two or more prior mitral valve surgeries.
What is the main result?
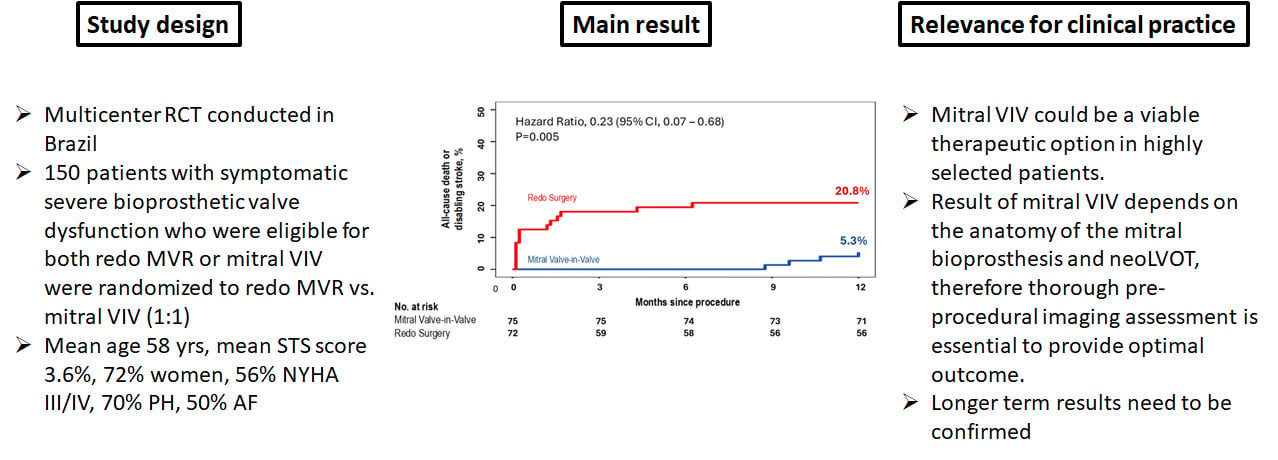
- At one year, the primary endpoint occurred in 20.8% of patients assigned to rMVR and in 5.3% of those treated with mVIV (hazard ratio 0.23 [95% CI 0.07-0.68], p=0.005). This difference was largely driven by events occurring in the early postoperative period.
- Mortality at 30 days was higher in the surgical group (12.5% vs. 0%). Life-threatening/major bleeding complications and acute kidney injury were also more frequently observed after surgery, while stroke rates were low in both groups.
- At one year, rehospitalisation rate due to cardiac causes was higher in the mVIV group (16% vs. 2.8%, p=0.02), including leaflet thrombosis event. Mean mitral gradient was lower (5.4 vs. 6.7mmHg, p=0.007) and mean prosthetic valve area was higher (1.7 vs. 1.4cm2, p=0.003) in the rMVR group.
Critical reading and the relevance for clinical practice
The SURVIV trial is the first RCT to directly compare mitral VIV vs. redo surgery in patients with symptomatic severe bioprosthetic valve dysfunction who are suitable for both redo MVR and mVIV. It is an important study to demonstrate that mVIV could be considered as a therapeutic option in this cohort. However, this trial result should be interpreted with caution, as it also has several limitations.
- Small trial (75 patients in each arm) within a single country, which could limit generalizability.
- Relatively young population (mean age 58 years) compared to Western cohorts (the average age for a first mitral valve surgery is about 64 years old in the US)
- 25% of patients had a history of two or more prior mitral valve surgeries and additional procedure was possible for surgical group (26% concomitant LAA closure, 14% concomitant tricuspid repair or replacement) which could have impacted the result
- No data are available on long-term outcome and valve durability (10 year follow-up planned)
Nevertheless, the SURVIV trial is the very first important RCT to demonstrate that early superiority of mitral VIV to redo MVR with respect to a composite of death or disabling stroke at 1 year, with a favourable safety profile among patients with symptomatic severe bioprosthetic valve dysfunction. These results suggest mVIV may offer an important short-term clinical benefit in highly selected patients.




No comments yet!