96 results
TRISCEND II: Two-year outcomes of transcatheter tricuspid valve replacement for severe tricuspid regurgitation
31 Mar 2026
Jonathan Curio provides his take on TRISCEND II trial results presented by Vinod H. Thourani at ACC.26 in New Orleans.

Author

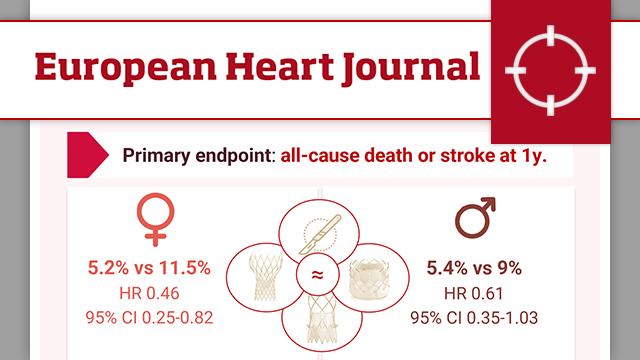
Sex-specific outcomes after transcatheter or surgical treatment of aortic valve stenosis: the DEDICATE-DZHK6 trial
30 Mar 2026
Women have long been underrepresented in TAVR trials. This article explores the DEDICATE trial’s sex-specific outcomes after TAVI and SAVR, revealing differences in early stroke risk, procedural challenges, and one-year survival in men and women with aortic stenosis.

Reviewer
ALL-RISE: A large-scale, global randomized trial of coronary physiology derived from conventional angiography compared with an invasive pressure wire-based approach to guide PCI
28 Mar 2026
Mirvat Al Asnag provides her take on the ALL-RISE randomised trial presented by Ajay J. Kirtane at ACC.26 in New Orleans.

Author

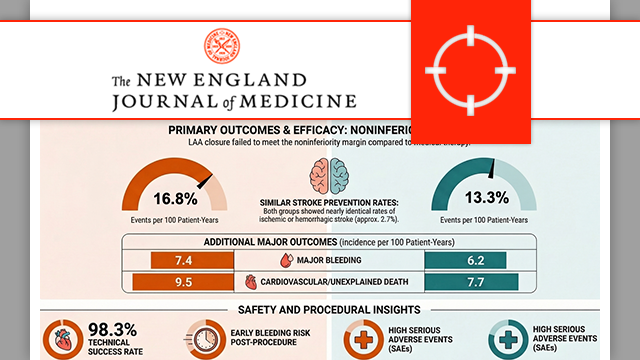
Outcomes in patients with atrial fibrillation randomized to receive LAA closure or oral anticoagulation: Primary results of the CHAMPION-AF clinical trial
28 Mar 2026
During the first Late-Breaking Trial session of the 2026 ACC Congress held in New Orleans, Saibal Kar, principal investigator of the CHAMPION-AF trial, presented the 3-year results of this groundbreaking study, which were simultaneously published in the NEJM (DOI: 10.1056/NEJMoa2517213).

Author

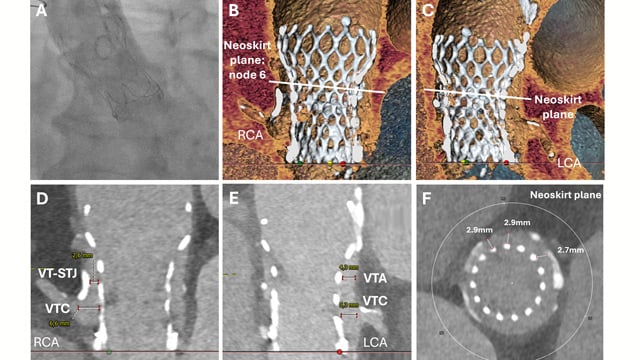
Rapid CT-guided emergency TAVI-in-TAVI-in-valve for cardiogenic shock with high coronary obstruction risk
01 Apr 2026
An 85-year-old man presented in cardiogenic shock with torrential aortic regurgitation after prior surgical and TAVI valve-in-valve procedures. With high coronary obstruction risk, urgent CT assessment and in-silico simulations guided a rapid redo TAVI-in-TAVI-in-valve implantation, demonstrating that CT-assisted emergency redo-TAVI is feasible and lifesaving in high-risk...
IVUS-guided versus angiography-guided PCI in unprotected left main coronary artery disease – The OPTIMAL trial
31 Mar 2026
Ali Nazmi Calik provides his take on the results of the OPTIMAL trial presented by Luca Testa at ACC.26 in New Orleans.

Author

Two-year outcomes after transcatheter tricuspid repair without cross-over in the randomised TRI-FR trial
28 Mar 2026
Alex Sticchi interviews Erwan Donal on the 2-year outcomes of the TRI-FR randomised trial evaluating transcatheter tricuspid edge-to-edge repair (T-TEER) versus medical therapy alone, as presented at ACC.26 in New Orleans.

Author

Author

Controlled trial of high-risk coronary intervention with percutaneous left ventricular unloading (CHIP-BCIS3)
01 Apr 2026
Aaysha Cader interviews Divaka Perera on the CHIP-BCIS3 randomised trial, which evaluated whether elective left ventricular (LV) unloading during high-risk PCI improves outcomes in patients with severe LV dysfunction, extensive coronary disease, and complex procedures, as presented at ACC.26 in New Orleans.

Author

Author

HOST-EXAM:10-year follow-up of clopidogrel vs aspirin monotherapy in stable CAD after PCI with drug-eluting stent
30 Mar 2026
Nicola Ryan provides her take on the 10-year follow-up of the HOST-EXAM, which was presented by Hyo-Soo Kim at ACC.26 in New Orleans.

Author

CLOSURE-AF: left atrial appendage closure or medical therapy in atrial fibrillation
23 Mar 2026
Catheter-based left atrial appendage closure (LAAC) is widely used as an alternative to oral anticoagulation in atrial fibrillation. But how does it compare with best medical therapy in patients at high risk of both stroke and bleeding? The CLOSURE-AF trial provides new data to inform this...

Reviewer