TRISCEND II: Two-year outcomes of transcatheter tricuspid valve replacement for severe tricuspid regurgitation
Reported from ACC.26
Jonathan Curio provides his take on TRISCEND II trial results presented by Vinod H. Thourani at ACC.26 in New Orleans.
Designed by Jonathan Curio. Source: PCRonline.com
Why this study? - the rationale/objective
Severe symptomatic tricuspid regurgitation (TR) has been identified as associated with increased morbidity and mortality. Tricuspid transcatheter edge-to-edge repair (T-TEER) has been established as very safe treatment option, providing patients with a significant symptomatic benefit (1). However, in a relevant proportion of patients T-TEER can provide only sub-optimal results in terms of residual TR, linked with worse clinical outcomes.
Here, transcatheter tricuspid valve replacement (TTVR) bears the promise to effectively eliminate all TR, ideally inducing a more pronounced impact on the patients’ clinical course. Against this background, the randomised TRISCEND II trial studied the EVOQUE TTVR system (Edwards Lifesciences) in comparison to optimised medical management only in patients with severe symptomatic TR. According to the 1-year results of 400 randomised patients, TTVR could very effectively reduce TR (TR ≤ mild in 95.3% at 1-year), and according to a primary composite endpoint, evaluated using a win-ratio approach, was superior to medical therapy alone, mainly driven by marked improvements in quality of life (2). However, according to the safety events analysis, severe bleeding was seen in 15.4%, and a new pacemaker implantation was needed in 17.4% of the TTVR patients.
Following these initial findings, the 2-year results of the TRISCEND II trial were eagerly awaited and have now been presented at this year’s ACC Congress (3).
How was it executed? – the methodology
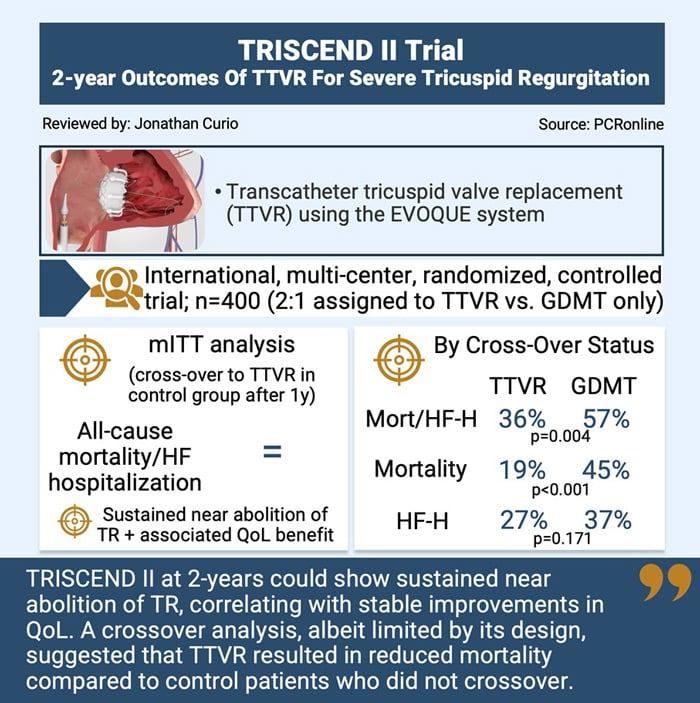
The TRISCEND II trial was an international, multi-centre, randomised, controlled trial that enrolled 400 patients with symptomatic severe TR, who were randomly assigned in a 2:1 ratio to either be treated with TTVR using the EVOQUE system in addition to medical therapy or to be treated with medical therapy alone. Relevant exclusion criteria were anatomical infeasibility to undergo TTVR, severely depressed right heart function, and severe pulmonary hypertension.
Endpoints of the trial included:
- Primary outcomes were a combination of all-cause mortality and heart-failure hospitalisation; these were also analysed separately
- Secondary endpoints of interest were the observed TR grade on follow-up, as well as quality of life measured per KCCQ-score
After 1 year, patients in the control group were allowed to cross over to be treated with TTVR. Thus, in the presented data in the intention-to-treat analysis a relevant effect of this cross-over has to be noted, while explorative analyses using the remaining small group of patients who continued on medical therapy only without cross-over were also conducted.
What is the main result?
The mean age of the patients was 79 years, and around 75% were female. 73% of patients in the TTVR group and 69% in the control group were in NYHA class III or IV, atrial fibrillation was present in over 90% of the patients, TR was of secondary etiology in 74% of the TTVR and in 71% of the control group patients.
- Assessing the study population as per intention-to-treat analysis (including those patients who crossed over to TTVR in the control group), at 2 years, there was no difference according to the combined endpoint of all-cause mortality or heart-failure hospitalisation
- However, assessing the study population by cross-over status, the endpoint was significantly reduced in those patients who received TTVR compared to those who received medical therapy only (36.0% vs. 57.4%, p = 0.004), which was driven be a reduced mortality rate at 2-years in the TTVR group (19.1% vs. 44.9%, p < 0.001); a Bayesian multiple imputation modeling analysis to estimate outcomes had the control group been preserved could confirm this significant reduction of all-cause mortality in the TTVR group (19.1% vs. 29.4%, p = 0.03)
- TR remained almost eliminated, with 95% ≤ mild TR at 2-years, and symptomatic benefits, as per increased KCCQ score, remained stable throughout continued follow-up
- In those patients of the medically managed control group that crossed over to TTVR after 1-year, also with delayed treatment a similarly effective TR reduction (95% ≤ mild TR), and equal symptomatic benefits were achievable
Critical reading and the relevance for clinical practice
The 2-year results of the TRISCEND II trial provide important additional insights regarding continued, longer-term follow-up of patients treated for severe symptomatic TR, in this case by means of TTVR.
The initial 1-year results for T-TEER, according to the randomised TRILUMINATE trial and the randomised Tri.Fr trial, as well as for TTVR in the TRISCIEND II trial, each comparing catheter-based intervention with medical therapy alone, could show relevant reduction in TR and significant quality of life improvements (1,2,4). However, in all these trials, the rates of hard endpoints, namely all-cause death and heart-failure hospitalisations, were without relevant difference between groups at 1-year follow-up.
Recently published 2-year data from the TRILUMINATE trial and the respective longer follow-up of the Tri.Fr trial, also presented at this year’s ACC congress, could show a significantly reduced rate of heart-failure hospitalisations in the T-TEER groups, beyond mere symptomatic improvements (5,6). The now presented 2-year follow-up of the TRISCEND II trial, in line with these noted findings for T-TEER, for the first time could establish a significant impact of TTVR on hard clinical endpoints, namely on all-cause mortality. Albeit the analysis was hampered by the crossovers that were allowed to be treated with TTVR in the medically managed control group, the clinical effect not only was seen in the analysis by crossover status (comparing the initial TTVR group with the relatively small group of patients who did not crossover from medical treatment until 2-years), but also in a post-hoc sensitivity analysis using Bayesian multiple imputation modeling to estimate outcomes had the control group been preserved, aiming to add an additional level of statistical rigor.
These 2-year findings of significantly reduced hard clinical endpoints seem to suggest that, indeed, the clinical perception of a vicious heart-failure cycle with constant volume-overload driven by severe TR can be ended effectively when TR is substantially reduced or even eliminated.
Of note, it seems important to actually bring down the degree of TR to mild or less, in order to enable a meaningful clinical benefit. Here, TTVR, in comparison to T-TEER seems to offer a relevant benefit, as in almost all cases near TR abolition seems achievable. On the contrary, T-TEER is often less predictable and more often results in relevant residual TR. However, this needs to be balanced against the excellent risk profile of T-TEER, with only very few complications noted in clinical trials as well as in real-world practice. TTVR, on the other hand, seems to be burdened by a notable rate of major bleedings and new pacemaker implantation associated with the procedure.
Taking these considerations together, it seems that the promise that apparently lies in transcatheter tricuspid valve interventions, now demonstrated by reduced hard clinical endpoints in T-TEER as well as TTVR trials, comes with the responsibility for multidisciplinary heart-teams to carefully select the adequate treatment modality for each individual patient, balancing the chances to achieve a meaningful TR reduction with potential risks of the chosen procedure.
In summary, the 2-year results of the TRISCEND II trial could show sustained near elimination of TR, correlating with stable improvements in quality of life. A crossover analysis, albeit limited by its design, suggested that TTVR resulted in reduced mortality compared to control patients who did not crossover.
References
- Sorajja P, Whisenant B, Hamid N, Naik H, Makkar R, Tadros P et al. Transcatheter Repair for Patients with Tricuspid Regurgitation. N Engl J Med 2023;Online ahead of print.
- Hahn RT, Makkar R, Thourani VH, Makar M, Sharma RP, Haeffele C et al. Transcatheter Valve Replacement in Severe Tricuspid Regurgitation. N Engl J Med 2025;392:115-126.
- Thourani V. Two-Year Outcomes of Transcatheter Tricuspid Valve Replacement for Severe Tricuspid Regurgitation: The TRISCEND II Randomized Trial. Presented at American College of Cardiology (ACC) Scientific Session, New Orleans, USA 2026.
- Donal E, Dreyfus J, Leurent G, Coisne A, Leroux PY, Ganivet A et al. Transcatheter Edge-to-Edge Repair for Severe Isolated Tricuspid Regurgitation: The Tri.Fr Randomized Clinical Trial. JAMA 2025;333:124-132.
- Kar S, Makkar RR, Whisenant BK, Hamid N, Naik H, Tadros P et al. Two-Year Outcomes of Transcatheter Edge-to-Edge Repair for Severe Tricuspid Regurgitation: The TRILUMINATE Pivotal Randomized Controlled Trial. Circulation 2025;151:1630-1638.
- Donal E. Two-year Outcomes After Transcatheter Tricuspid Repair Without Cross-over In The Randomized Tri-fr Trial. Presented at American College of Cardiology (ACC) Scientific Session, New Orleans, USA 2026.





No comments yet!