Transcatheter aortic valve implantation without routine percutaneous coronary intervention: A randomized controlled trial (PRO-TAVI Trial)
Reported from ACC.26
Jonathan Curio provides his take on the PRO-TAVI trial presented by Michiel Voskuil at ACC.26 in New Orleans and simultaneously published in The Lancet.
Designed by Jonathan Curio. Source: PCRonline.com
Why this study? - the rationale/objective
Transcatheter aortic valve implantation (TAVI) has fundamentally transformed the treatment of patients with severe aortic stenosis (AS). With positive data from large, randomized trials across the whole surgical risk spectrum, according to recent guidelines it has become the preferred treatment option, especially in patients ≥ 70 years of age (1).
Still, several aspects of optimized patient care and procedural strategy need further study. In particular, the management of concomitant stable coronary artery disease (CAD), present in around 50% of patients undergoing TAVI, is an ongoing matter of debate (2). Several studies could not demonstrate a clear clinical benefit when treating concomitant relevant obstructive CAD.
Thus, the PRO-TAVI trial, which simultaneously to presentation at the ACC congress was published in the Lancet, aimed to evaluate whether deferral of routine percutaneous coronary intervention (PCI) in patients undergoing TAVI is non-inferior in comparison to TAVI with preceding PCI (3).
How was it executed? – the methodology
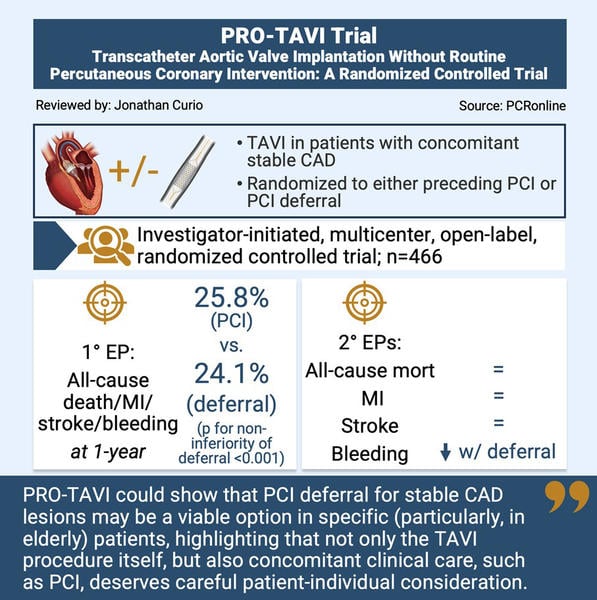
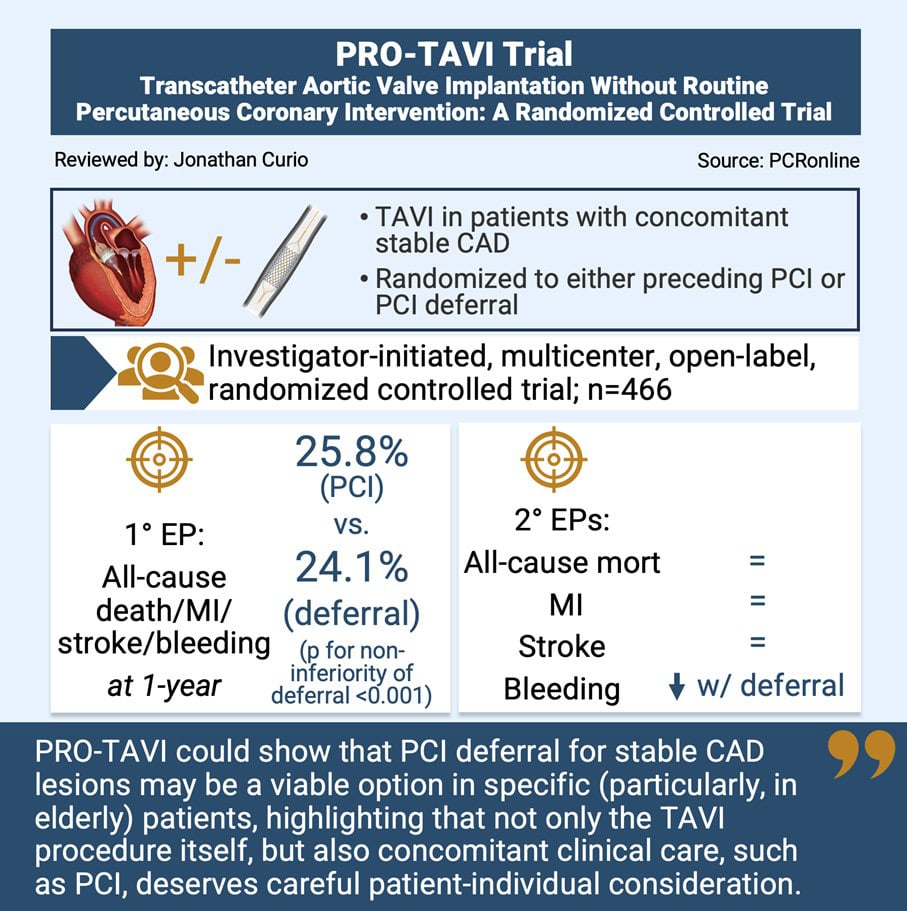
The PRO-TAVI trial was an investigator-initiated, multicenter, open-label, randomized controlled trial enrolling patients who underwent TAVI and presented with relevant concomitant stable CAD. Patients were included if the multidisciplinary heart team had decided to treat them by means of TAVI and if concomitant CAD, defined as a) a coronary stenosis of 70% to 99% or b) an intermediate stenosis of 40% to 70% combined with positive physiological parameters was present. Exclusion criteria were an unprotected left main stenosis, no PCI-eligible stenoses, or a contraindication for DAPT.
The patients were randomised in a 1:1 fashion to either routine PCI before TAVI or PCI deferral.
Endpoints of the trial included:
- Primary endpoint: 1-year composite of all-cause death, myocardial infarction, stroke, or major bleeding
- Secondary endpoints: Individual components of the primary endpoint, as well as a revascularisation analysis
What is the main result?
The trial enrolled 466 patients. Median age of the trial population was 81 years, and 35% of patients in the PCI group and 36% in the deferral group were female. The STS risk-score for operative mortality was 3.1%. The median SYNTAX score of the present lesions was 10, of the lesions 21% were proximal LAD, and physiological assessment had been performed in 14%, each equivalent for both groups. TAVI was performed via transfemoral access in 93% of cases in both groups, and in both groups around half of the patients received a balloon-expandable and half received a self-expandable device.
- According to the combined primary endpoint at 1 year, there was no significant difference between performed PCI and PCI deferral (p for noninferiority of deferral <0.001, p for superiority = 0.68)
- Assessing the individual components of the primary endpoint there was no difference between groups for the incidence of death from any cause, myocardial infarction, and stroke, however, at 1-year there was a notably higher rate of relevant bleedings in the group of patients with PCI as compared to those with PCI deferral (6.2% vs. 14.8%, HR: 0.39, 95% CI: 0.21-0.73)
- During follow-up, patients in the deferral group received more revascularisation procedures (10.8% vs. 4.8%) with a median time between randomisation and the procedure of 87 days, however, there was no relevant difference in the number of urgent revascularisations, and all PCI procedures after TAVI could be performed without any major periprocedural complications
Critical reading and the relevance for clinical practice
The results of the PRO-TAVI trial mean an important addition to the ever-growing body of evidence regarding optimal care of patients with severe AS, and in this case concomitant stable CAD.
According to the findings of the trial, a strategy of PCI deferral, could be non-inferior to PCI performed in the run-up of a scheduled TAVI procedure, and such strategy may result in less major bleedings during the 1-year follow-up period.
Before, two other relevant randomised trials aimed to shed light on this important clinical question. The ACTIVATION trial testing a strategy of PCI before TAVI had to be stopped due to futility and could not show non-inferiority of PCI at 1-year, while noticing a higher rate of bleeding in the PCI group (4). The more recent NOTION-3 trial showed a reduced incidence of its combined primary endpoint of all-cause mortality, myocardial infarction, and urgent revascularization at a median follow-up of 2-years (5). Of note, this benefit was most pronounced in patients with lesions of ≥ 90% angiographic severity. However, also in NOTION-3 there was a signal of a higher bleeding rate in the PCI group. In line with this, non-inferiority in the present PRO-TAVI trial was mainly driven by a lower rate of major bleedings, while PCI deferral was associated with a numerically higher incidence of the combined merely ischemic endpoint of all-cause mortality, myocardial infarction, and stroke. Here, the combination of ischemic and bleeding events in the composite primary endpoint may need to be discussed as a limitation of the PRO-TAVI trial.
Still, these findings may encourage choosing a strategy of PCI deferral until actual PCI is clinically indicated. Indeed, only around 10% of the patients in the PCI deferral group had to undergo a PCI procedure during follow-up, due to continued symptoms and these procedures were easily feasible with only few complications noted. In line with this, recent 2025 ESC/EACTS guidelines give the treatment of stenoses ≥ 90% a class IIa recommendation, with a lower recommendation to treat stenoses of only 70%-90% - given the PRO-TAVI data deferral of any of those lesions may be reasonable, potentially sparing invasive procedures, possible complications, and health-care costs.
Of note, both in the present PRO-TAVI trial with a median SYNTAX score of 10 as well as in NOTION-3 with a mean SYNTAX score of 9 the significance of CAD was modest and relevant potential lesions, namely unprotected left main stenoses, were excluded. Furthermore, the treated cohort was rather elderly, thus, the results may not be fully applicable to younger patients. This needs to be considered when thinking about the generalizability of the trial results and considering how to implement them in actual clinical practice.
Taking all these aspects together, it becomes evident that contemporary execution of TAVI procedures requires a patient-tailored therapy not only regarding the actual valve implantation (e.g., device choice, implant position, alignment, considering coronary access as well as TAVI ReDo feasibility), but also regarding concomitant clinical factors such as stable CAD lesions. Heart team members need to perform a detailed analysis of the lesion at hand and balance ischemic and bleeding risks of the individual patient.
In conclusion, the PRO-TAVI trial could show that a strategy of PCI deferral for stable CAD lesions in specific (particularly in elderly) patients undergoing TAVI may be a viable option, highlighting that not only the TAVI procedure itself, but also concomitant clinical care, such as PCI, deserves careful patient-individual consideration.
References
- Praz F, Borger MA, Lanz J, Marin-Cuartas M, Abreu A, Adamo M et al. 2025 ESC/EACTS Guidelines for the management of valvular heart disease. Eur Heart J 2025;46:4635-4736.
- Tomii D, Lanz J, Thiele H, Heg D, Kim WK, Joner M et al. Obstructive Coronary Artery Disease and Health Status in Transcatheter Aortic Valve Replacement: A Post Hoc Analysis of the SCOPE I Randomized Clinical Trial. JAMA Netw Open 2025;8:e2547111.
- Delewi R, Aarts HM, Broeze GM, Hemelrijk KI, van Ginkel DJ, Versteeg GAA et al. Deferral of percutaneous coronary intervention in patients undergoing transcatheter aortic valve implantation (PRO-TAVI): an investigator-initiated, multicentre, open-label, non-inferiority, randomised controlled trial. Lancet 2026.
- Patterson T, Clayton T, Dodd M, Khawaja Z, Morice MC, Wilson K et al. ACTIVATION (PercutAneous Coronary inTervention prIor to transcatheter aortic VAlve implantaTION): A Randomized Clinical Trial. JACC Cardiovasc Interv 2021;14:1965-1974.
- Lonborg J, Jabbari R, Sabbah M, Veien KT, Niemela M, Freeman P et al. PCI in Patients Undergoing Transcatheter Aortic-Valve Implantation. N Engl J Med 2024;391:2189-2200.





No comments yet!