Aspirin discontinuation timing with ticagrelor monotherapy in ACS: a post hoc analysis from TICO and T-PASS
Selected in EuroIntervention Journal by N. Ryan
The TICO1 and T-PASS2 trials evaluated abbreviated DAPT followed by ticagrelor monotherapy compared to DAPT in patients with ACS undergoing PCI. Both trials demonstrated the safety and efficacy of abbreviated DAPT however, the optimal timing of aspirin discontinuation remains uncertain. This post hoc analysis compares the safety and efficacy of aspirin discontinuation at less than one month (T-PASS) to aspirin discontinuation at three months (TICO) post PCI for ACS.
References
Authors
Jung-Hee Lee, Jaeoh Lee, Su Yong Kim, Ho Sung Jeon, Jun-Won Lee, Sung Gyun Ahn, Yong-Joon Lee, Seung-Jun Lee, Chul-Min Ahn, Jung-Sun Kim, Byeong-Keuk Kim, Young-Guk Ko, Donghoon Choi, Myeong-Ki Hong, Yangsoo Jang, Sung-Jin Hong, and Young Jin Youn
Reference
2026;22:232-242 DOI: 10.4244/EIJ-D-25-00832
Published
16 February 2026
Link
Read the abstractReviewer
My Comment
Why this study – the rationale/objective?
Antiplatelet therapy post PCI aims to reduce ischaemic events however, comes with the concomitant increased risk of bleeding. Traditionally, following a period of dual antiplatelet therapy, aspirin monotherapy is prescribed lifelong to reduce the ischaemic risk. More recently trials3-5 have suggested the P2Y12 monotherapy may be preferable in terms of reducing bleeding risk without increasing ischaemic risk. Both TICO and T-PASS demonstrated a reduction in bleeding events without an ischaemic penalty in patients treated with short-duration DAPT followed by ticagrelor monotherapy at one-year. However aspirin was discontinued at different time-frames therefore the authors attempted to compare the safety and efficacy of aspirin discontinuation at one month to aspirin discontinuation at three months post PCI for ACS.
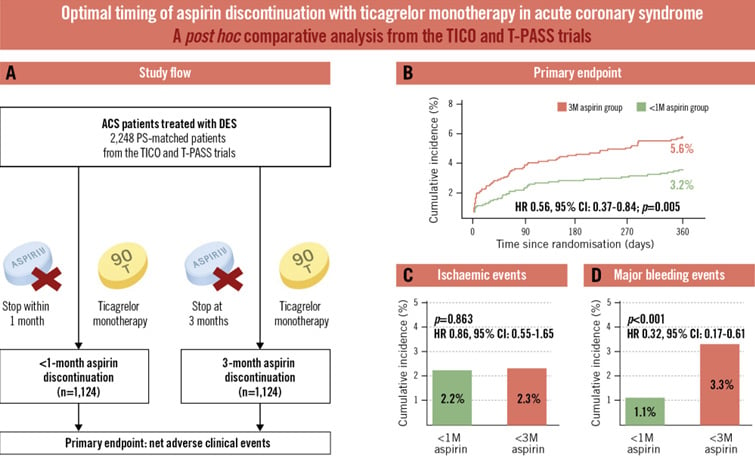
Central illustration. Aspirin discontinuation within 1 month versus at 3 months with ticagrelor monotherapy in acute coronary syndrome. A) Study flow. Cumulative incidences of the primary endpoint (B), ischaemic events (C), and major bleeding events (D). 1M: 1 month; 3M: 3 months; ACS: acute coronary syndrome; CI: confidence interval; DES: drug-eluting stent; HR: hazard ratio; PS: propensity score; T-PASS: Ticagrelor Monotherapy in PAtients Treated With New-generation Drug-eluting Stents for Acute Coronary Syndrome; TICO: Ticagrelor Monotherapy After 3 Months in the Patients Treated With New Generation Sirolimus Stent for Acute Coronary Syndrome
Source: EuroIntervention Journal
How was it executed? The methodology
The TICO and T-PASS datasets were combined and patients randomised to 12 months DAPT were excluded leaving 2,953 patients who received abbreviated DAPT followed by ticagrelor monotherapy in this analysis. Both trials were multicentre carried out within the Republic of Korea and patients were treated with bioresorbable polymer sirolimus eluting stents. Overall compliance to assigned treatment was 88% in TICO and 86% in T-PASS.
- The primary endpoint was net adverse clinical events (NACE); a composite of all-cause mortality, myocardial infarction, stent thrombosis, ischaemia-driven target vessel revascularisation, stroke, and major bleeding within one year.
- Secondary endpoints were the individual components of the primary endpoint, and MACCE, a composite of all-cause death, myocardial infarction, stent thrombosis, ischaemia-driven target vessel revascularisation and stroke.
What is the main result?
Recruitment occurred between August 2015 and October 2018 for TICO with 1,527 patients assigned to aspirin discontinuation at three months. In the T-PASS trial recruitment occurred between April 2019 and May 2022 with 1,426 patients assigned to <1 month aspirin, in this group aspirin was discontinued at a median of 16 days. After propensity score matching, there were 1,124 patients in each group with no significant differences in clinical, lesion and procedural characteristics between groups. The majority of patients were male in their 60's with 38% presenting with STEMI.
- NACE occurred less frequently in the <1 month aspirin group compared to 3 months aspirin at one year (3.2% vs. 5.6%, HR 0.56, 95%CI 0.37-0.84, p=0.005).
- There was no difference in MACE between groups at one year (2.2% vs. 2.3%, HR 0.86, 95%CI 0.55-1.65, p=0.863).
- Major bleeding was lower in the <1 month aspirin group compared to 3 months aspirin at one year (1.1% vs 3.3%, HR 0.32, 95%CI 0.17-0.61, p<0.001).
- In a three-month landmark analysis
- NACE rates diverged with the first 90 days (HR 0.55, 95%CI 0.32-0.94, p=0.013).
- There was no significant heterogeneity in the treatment effects between the early and late periods (p for interaction = 0.79).
Critical reading and the relevance for clinical practice:
The results of this study show that at 1-year ticagrelor monotherapy after <1 month of DAPT lead to improved clinical outcomes compared to aspirin discontinuation at three months in terms of NACE. The reduced NACE with aspirin discontinuation at <1 month was primarily driven by a reduction in bleeding events with no significant differences in MACCE between the groups.
The findings are similar to those of the STOPDAPT-2 trial5 where one-month DAPT followed by clopidogrel monotherapy was superior to one year DAPT in terms of NACE. Different to STOPDAPT-2, this analysis included only patients presenting with acute MI, whilst two-thirds of the STOPDAPT-2 population had chronic coronary syndrome. Both trials included in this analysis recruited in the Republic of Korea, whilst STOPDAPT-2 recruited in Japan. It is recognised East Asian populations have a higher risk of major bleeding compared to a Western population therefore, these results may not be generalisable. However, the TARGET-FIRST8 trial which was undertaken in European centres demonstrated that 1-month DAPT followed by P2Y12 inhibitor monotherapy was non-inferior to DAPT in terms of a composite of death from any cause, myocardial infarction, stent thrombosis, stroke, or major bleeding with lower bleeding events. Importantly the TARGET-FIRST trial included low-risk MI patients who had undergone complete revascularisation.
The optimal time to discontinue aspirin and continue P2Y12 inhibition alone is not clear particularly given the results of NEO-MINDSET6 which failed to demonstrate non-inferiority of potent P2Y12 inhibition alone within the first 4 days post PCI compared to DAPT in patients undergoing PCI for ACS. In the STOP-DAPT 37 comparing prasugrel alone compared to DAPT was non-inferior in terms of cardiovascular events at one month post PCI however importantly failed to demonstrate superiority in terms of reduced bleeding risk with a signal suggestive of excess coronary events in the prasugrel groups.
As with any analysis, there are a number of limitations which must be borne in mind most importantly whilst the individual trials were randomised this is a post-hoc analysis therefore, all findings are observational in nature. The trials utilised a single stent platform and whilst propensity matching was carried out the inclusion criteria differed between the trials which could potentially lead to unmeasured confounders.
This study adds to the data with regard to the anti-platelet strategy in the ACS population, it would appear that the one-month point is a good opportunity to reassess the bleeding and ischaemic risk. Ongoing trials evaluating the immediate discontinuation of aspirin LEGACY (NCT05125276) will add to the evidence. To date the optimal duration of DAPT and longer-term antiplatelet strategy in all populations remains undetermined and clinicians must carefully assess individual patients bleeding and ischaemic risks when prescribing and reviewing antiplatelet therapy.
References
- Kim BK, Hong SJ, Cho YH, et al. Effect of Ticagrelor Monotherapy vs Ticagrelor With Aspirin on Major Bleeding and Cardiovascular Events in Patients With Acute Coronary Syndrome: The TICO Randomized Clinical Trial. JAMA. 2020;323(23):2407–2416.
- Stopping Aspirin Within 1 Month After Stenting for Ticagrelor Monotherapy in Acute Coronary Syndrome: The T-PASS Randomized Noninferiority Trial. Circulation. 2024.
- Koo BK, Kang J, Park KW, et al. Aspirin versus clopidogrel for chronic maintenance monotherapy after percutaneous coronary intervention (HOST-EXAM). Lancet. 2021.
- Baber U, Dangas G, Angiolillo DJ, et al. Ticagrelor alone vs. ticagrelor plus aspirin following percutaneous coronary intervention in patients with non-ST-segment elevation acute coronary syndromes: TWILIGHT-ACS. Eur Heart J. 2020;41(37):3533–3545.
- Watanabe H, Domei T, Morimoto T, et al. Effect of 1-Month Dual Antiplatelet Therapy Followed by Clopidogrel vs 12-Month Dual Antiplatelet Therapy on Cardiovascular and Bleeding Events in Patients Receiving PCI: The STOPDAPT-2 Randomized Clinical Trial. JAMA. 2019;321(24):2414.
- Guimarães PO, Franken M, Tavares CAM, et al. Early Withdrawal of Aspirin after PCI in Acute Coronary Syndromes. N Engl J Med. 2025;393(21):2095–2106.
- Natsuaki M, Watanabe H, Morimoto T, et al. An Aspirin-Free Versus Dual Antiplatelet Strategy for Coronary Stenting: STOPDAPT-3 Randomized Trial. Circulation. 2024;149(8):585–600.
- Tarantini G, Honton B, Paradies V, et al. Early Discontinuation of Aspirin after PCI in Low-Risk Acute Myocardial Infarction. N Engl J Med. 2025;393(21):2083–2094.




No comments yet!