IVUS-guided versus angiography-guided PCI in unprotected left main coronary artery disease – The OPTIMAL trial
Reported from ACC.26
Ali Nazmi Calik provides his take on the results of the OPTIMAL trial presented by Adrian Banning at ACC.26 in New Orleans.
Designed by Ali Nazmi Calik. Source: PCRonline.com
Why this study – the rationale/objective?
Percutaneous coronary intervention (PCI) has become an established alternative to coronary artery bypass grafting for patients with unprotected left main coronary artery (LMCA) disease, particularly in those with low-to-intermediate anatomical complexity. However, LMCA PCI remains technically demanding, requiring optimal stent expansion, apposition, and lesion coverage that are key determinants of long-term outcomes.
Intravascular ultrasonography (IVUS) offers high-resolution intraluminal imaging that can guide stent sizing and optimisation. Observational studies and prior randomised trials in broader PCI populations have suggested that IVUS guidance reduces adverse events, including mortality, myocardial infarction, and stent thrombosis. Consequently, contemporary guidelines strongly recommend intracoronary imaging, especially for complex lesions such as left main disease. Despite this, robust randomised evidence specifically focused on unprotected LMCA PCI was lacking, and existing data were largely extrapolated from heterogeneous populations.
The OPTIMAL trial was therefore designed to determine whether routine IVUS-guided PCI improves clinical outcomes compared with angiography-guided PCI in patients with unprotected LMCA disease.
How was it executed - the methodology?
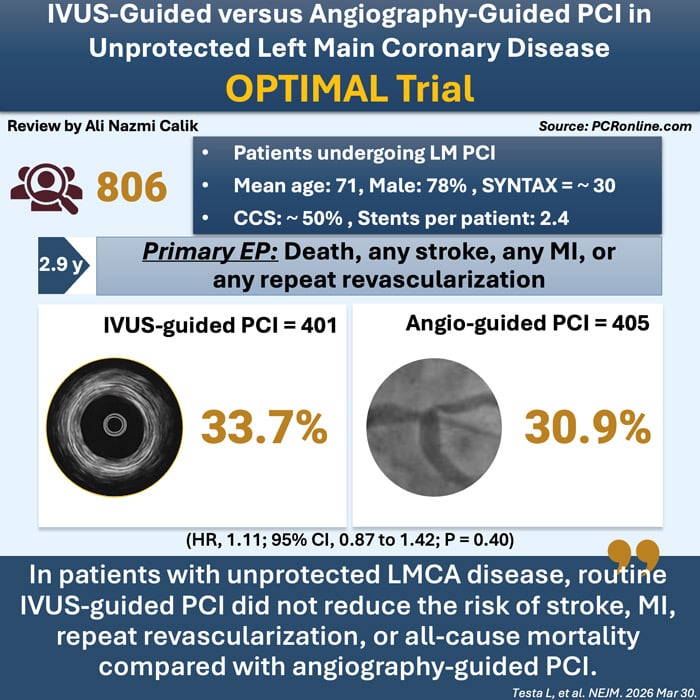
The OPTIMAL trial was an investigator-initiated, international, multicenter, randomised, open-label superiority trial conducted across 28 high-volume European centres. A total of 806 patients with LMCA disease, defined as a stenosis of at least 50% and deemed suitable for PCI, were enrolled. The study population reflects contemporary real-world practice, with a mean age of 71 years and an intermediate-to-high anatomical complexity, as indicated by a mean SYNTAX score of approximately 30.
Patients were randomly assigned in a 1:1 ratio to undergo either IVUS-guided PCI or angiography-guided PCI. In the IVUS-guided group (n=401), pre-procedural imaging was strongly recommended, and post-procedural IVUS assessment was mandatory to ensure optimal stent deployment according to predefined criteria, including minimum lumen area thresholds such as ≥8 mm² in the left main body. In contrast, in the angiography-guided group (n=405), the use of IVUS was discouraged and permitted only in cases where it was deemed necessary for patient safety. In both groups, PCI was performed using contemporary techniques and second-generation drug-eluting stents.
The primary endpoint was a patient-oriented composite endpoint (PoCE), defined as the occurrence of all-cause death, any stroke, any myocardial infarction, or any repeat revascularisation.
The secondary end points included a device-oriented composite of death from cardiovascular causes, target-vessel myocardial infarction, or clinically indicated target-lesion revascularisation; a vessel-oriented composite of death from cardiovascular causes, target-vessel myocardial infarction, or target-vessel revascularisation; and death from any cause.
Patients were followed for a median duration of 2.9 years. The trial was designed and powered to test the superiority of IVUS guidance, based on the assumption of a substantial relative reduction in adverse clinical events.
What is the main result?
The OPTIMAL trial demonstrated a neutral result with respect to its primary endpoint. At a median follow-up of 2.9 years, the patient-oriented composite endpoint of death, myocardial infarction, stroke, or repeat revascularization occurred in 33.7% of patients in the IVUS-guided PCI group and in 30.9% of those in the angiography-guided PCI group, with a hazard ratio of 1.11 (95% CI 0.87–1.42; P=0.40), indicating no significant difference between the two strategies.
Importantly, there were no meaningful differences across the individual components of the composite endpoint. Rates of all-cause mortality were similar between groups (15.7% vs. 15.1%), as were rates of myocardial infarction (11.2% vs. 10.9%) and repeat revascularisation (12.0% vs. 11.1%). These findings suggest that IVUS guidance did not translate into a reduction in either hard clinical endpoints or repeat procedures in this population.
A notable and unexpected finding was a higher incidence of stroke in the IVUS group compared with the angiography group (3.0% vs. 1.0%; hazard ratio 3.11). However, these events occurred late, with a median time to stroke of 19 months, making a direct procedural relationship unlikely. Procedurally, IVUS guidance was associated with longer procedure times, but this did not result in an increase in periprocedural complications. Interestingly, IVUS led to additional optimisation manoeuvres such as dilation or balloon inflation after stent placement or additional stenting in only 29.3% of the cases.
Critical reading and the relevance for clinical practice
The present trial demonstrated that, in patients with unprotected left main coronary artery disease, routine IVUS-guided PCI did not reduce the risk of stroke, myocardial infarction, repeat revascularisation, or all-cause mortality compared with angiography-guided PCI over a median follow-up of 2.9 years.
The neutral findings of the OPTIMAL trial challenge the prevailing assumption that routine IVUS guidance is necessary for all left main PCI procedures and call for a more nuanced interpretation of its role in contemporary practice. One of the most important considerations is the level of operator expertise. The trial was conducted in high-volume centres by operators who were already highly experienced in IVUS use prior to study initiation. This likely resulted in a form of “angiographic recalibration,” whereby operators were able to achieve optimal stent deployment using angiographic guidance alone, informed by their prior intracoronary imaging experience. As a result, the difference between IVUS-guided and angiography-guided PCI may have been attenuated.
In addition, the overall quality of PCI was very high in both groups, with frequent use of post-dilatation, proximal optimisation techniques, and contemporary drug-eluting stents. This suggests that modern PCI practice may already incorporate many of the principles traditionally reinforced by IVUS, thereby reducing its incremental benefit. Supporting this interpretation, IVUS findings led to additional corrective measures in only approximately 30% of patients, emphasising that imaging itself is not therapeutic unless it meaningfully alters procedural strategy.
The IVUS optimization criteria used in the trial, targeting a minimum stent area of 8 mm² in the LM body, 7 mm² at the polygon of confluence (POC), 6 mm² in the LAD, and 5 mm² in the LCX, based on the criteria proposed by Kang et al. may be open to discussion, as more recent data suggest that larger MSA thresholds are associated with improved outcomes. However, the actual results of the study demonstrated that the achieved mean MSA values were substantially higher, with measurements exceeding 11 mm² in the distal LM, 8 mm² in the ostial LAD, and 6.5 mm² in the ostial LCX, aligning well with these more contemporary recommendations.
The observed excess of stroke in the IVUS group, although statistically significant, is difficult to interpret clinically. The delayed timing of these events argues against a causal procedural mechanism and raises the possibility of a chance finding, although it highlights the need for further investigation in future studies.
From a clinical perspective, the findings of this trial are unlikely to lead to major changes in current clinical practice regarding the use of IVUS in unprotected left main PCI. The key message of the OPTIMAL trial is that optimal procedural execution is more important than the routine use of any single adjunctive tool. In left main PCI, achieving adequate stent expansion and apposition remains the primary determinant of outcomes, regardless of whether this is guided by IVUS or by expert angiographic judgment.





No comments yet!