CLOSURE-AF: left atrial appendage closure or medical therapy in atrial fibrillation
Selected in The New England Journal of Medicine by S. Brugaletta
Catheter-based left atrial appendage closure (LAAC) is widely used as an alternative to oral anticoagulation in atrial fibrillation. But how does it compare with best medical therapy in patients at high risk of both stroke and bleeding? The CLOSURE-AF trial provides new data to inform this question.
References
Authors
Ulf Landmesser, Carsten Skurk, Paulus Kirchhof, Thorsten Lewalter, Johannes Hartung, Andi Rroku, Burkert Pieske, Johannes Brachmann, Ibrahim Akin, Claudius Jacobshagen, Benjamin Meder, Andreas Zeiher, Stefan D. Anker, Holger Thiele, Stefan Blankenberg, Steffen Massberg, Heribert Schunkert, Norbert Frey, Alexander Joost, Martin Bergmann, Ralph Stephan von Bardeleben, Tim Friede, Marius Placzek, Anna Suling, Karl Georg Haeusler, Matthias Endres, Karl Wegscheider, Leif-Hendrik Boldt, and Ingo Eitel, for the CLOSURE-AF Trial Investigators
Reference
DOI: 10.1056/NEJMoa2513310
Published
Published March 18, 2026
Link
Read the abstractReviewer
My Comment
CLOSURE-AF Trial at a glance: design, outcomes, and insights. Created with AI support by Salvatore Brugaletta for PCRonline -
Why this study – the rationale/objective?
Catheter-based closure of the left atrial appendage is an alternative to oral anticoagulation for stroke prevention in patients with atrial fibrillation.
The effectiveness of this strategy, as compared with physician-directed best medical care, in patients at high risk for stroke and bleeding is unknown.
How was it executed? The methodology
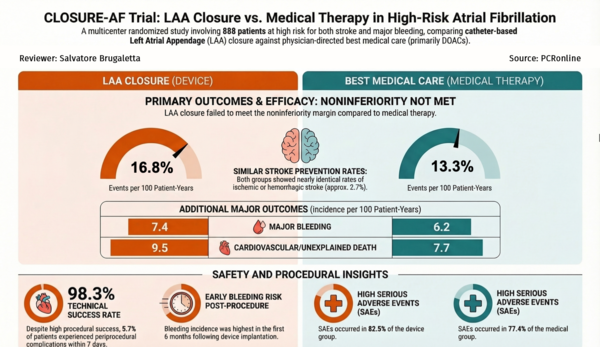
In this multicenter randomised trial conducted in Germany, patients with atrial fibrillation and a high risk of stroke and bleeding were randomised to undergo left atrial appendage closure or to receive physician-directed best medical care (including direct oral anticoagulants, if eligible).
The primary endpoint, tested for non-inferiority, was a composite of stroke (ischemic or hemorrhagic), systemic embolism, major bleeding, or cardiovascular or unexplained death, assessed in a time-to-event analysis. The non- inferiority margin was a hazard ratio of 1.3.
What is the main result?
A total of 912 adult patients underwent randomisation.
The primary endpoint analysis included 446 patients who were assigned to undergo left atrial appendage closure (device group), and 442 who were assigned to physician-directed best medical care (medical-therapy group). The mean CHA2DS2-VASc score was 5.2 ± 1.5, and the mean HAS-BLED score was 3.0 ± 0.9.
After a median follow-up of 3 years (interquartile range, 1.7 to 4.7), a first primary endpoint event had occurred in 155 patients (incidence per 100 patient-years, 16.8) in the device group, and in 127 patients (incidence per 100 patient-years, 13.3) in the medical-therapy group (difference in restricted mean survival time, −0.36 years; 95% confidence interval, −0.70 to −0.01; P = 0.44 for non-inferiority).
Serious adverse events occurred in 368 patients (82.5%) in the device group and 342 (77.4%) in the medical-therapy group.
Critical reading and the relevance for clinical practice:
Waiting for the results of the CHAMPION-AF, which will be presented at the ACC. 2026, the results of the CLOSURE-AF presented months ago at the AHA 2025 have been finally published. This gives us the possibility to see the details of the trial and to analyse the results.
The trial has been well executed from the methodological point of view. Its findings are quite disruptive and they go against a procedure - LAAC - which is largely performed. Analysing specifically all the various points of the trial, it is difficult to find out an explanation of these negative results, and this may give us a word of caution in performing large volume of LAAC, which is probably a procedure to be restricted to specific patients instead of being performed in any atrial fibrillation at high risk of ischemic and bleeding events.
An important point to be discussed is management of antiplatelet regimen after LAAC, which many experts may say was the cause of negativity of the trial and of the excess of bleeding. We should take in mind that the medical treatment group received mainly DOAC, which in theory may be even worse than dual antiplatelet therapy in terms of bleeding events.
The truth is that there is no a consensus about which antiplatelet regimen to use after LAAC.
Do this trial change your practice about LAAC?




No comments yet!