HOST-EXAM:10-year follow-up of clopidogrel vs aspirin monotherapy in stable CAD after PCI with drug-eluting stent
Reported from ACC.26
Nicola Ryan provides her take on the 10-year follow-up of the HOST-EXAM, which was presented by Hyo-Soo Kim at ACC.26 in New Orleans.
Designed by Nicola Ryan. Source: PCRonline.com
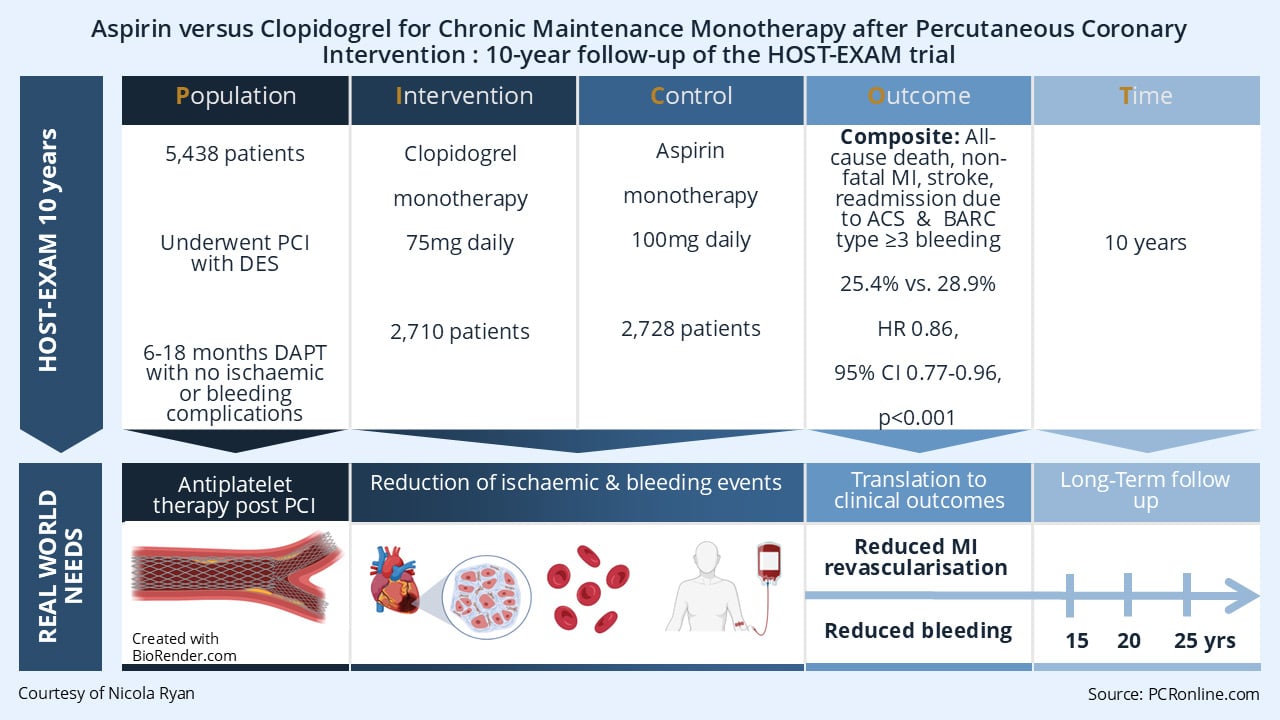
The HOST-EXAM trial (1) was a prospective randomised, open-label trial investigating clopidogrel monotherapy versus aspirin monotherapy in patients undergoing PCI with DES who had completed 6-18 months of DAPT with no ischaemic or bleeding complications. At 24 months follow-up, clopidogrel monotherapy significantly reduced the risk of the composite endpoint of all-cause death, non-fatal MI, stroke, readmission due to ACS and BARC 3-5 bleeding with previous extended follow-up to a median of 5.8 years, demonstrating this was a sustained benefit (2). Here, the authors report the 10-year follow-up to assess the very long-term effects of clopidogrel versus aspirin monotherapy.
Why this study – the rationale/objective?
Antiplatelet therapy post PCI aims to reduce ischaemic events however, comes with the concomitant increased risk of bleeding. Traditionally, following a period of dual antiplatelet therapy, aspirin monotherapy is prescribed lifelong to reduce the ischaemic risk. More recently, trials (1,3–7) have suggested the P2Y12 monotherapy may be preferable in terms of reducing bleeding risk without increasing ischaemic risk. In the HOST-EXAM trial at both the initial two-year follow-up as well as the initial extended follow-up, there was a significant reduction in both bleeding and ischaemic events in the clopidogrel arm. Given that antiplatelet therapy is typically prescribed lifelong, it is of interest to understand the longer-term outcomes in this population.
How was it executed - the methodology?
Patients undergoing PCI with a drug-eluting stent who completed 6-18 months of DAPT without any ischaemic or major bleeding complications were eligible for inclusion. Patients were randomised 1:1 to monotherapy with clopidogrel 75mg daily or aspirin 100mg daily. After the initial two-year trial follow-up period, the antiplatelet agent was at the discretion of the treating physician.
- The primary outcome was a composite of all-cause death, non-fatal MI, stroke, readmission due to acute coronary syndrome and BARC type ≥3 bleeding.
- The secondary composite endpoints included the thrombotic composite endpoint (cardiac death, non-fatal MI, ischaemic stroke, readmission due to ACS and definite or probable stent thrombosis) and any bleeding (BARC type ≥ 2).
- Individual components of the primary and secondary composite endpoints were included as secondary endpoints.
What is the main result?
Overall, between March 2014 and May 2018, 5,438 patients were included in the trial and in this intention-to-treat analysis. Overall, 2,138 patients in the clopidogrel monotherapy and 2,041 patients in the aspirin monotherapy group adhered to the protocol during the 10-year follow-up.
- The composite primary endpoint occurred in 25.4% of the clopidogrel group compared to 28.5% of the aspirin group. (HR 0.86, 95% CI 0.77-0.96, p<0.005). Giving an absolute risk reduction of 3.1 (95%CI 0.6-5.6), NNT 32.7.
- The thrombotic endpoint was lower in the clopidogrel group compared to the aspirin group. (17.3% vs. 20.0% HR 0.72, 95%CI 0.72-0.93 p<0.0024). Giving an absolute risk reduction of 2.8 (95%CI 0.6-5.0), NNT 35.9
- Similarly, any bleeding was lower with clopidogrel (9.1% vs 10.8% HR 0.81, 95% CI 0.68-0.97, p=0.02).
- There were no significant differences in all-cause death between the groups (clopidogrel 13.4% vs aspirin 12.5% HR 1.07, 95%CI 0.92-1.24, p=0.40).
- Landmark analysis showed the benefit of clopidogrel was consistent during both the initial trial period as well as the extended follow-up.
Critical reading and the relevance for clinical practice
The results of this extended follow-up of the HOST-EXAM trial show that the benefit of clopidogrel monotherapy in terms of reduction of both ischaemic and bleeding risk in patients who have tolerated 6-18 months of DAPT post PCI without ischaemic or haemorrhagic complications persists through longer-term follow-up.
The authors should be congratulated for the longest follow-up of SAPT post PCI, which is particularly important, given the ageing population, increased treatment with PCI and requirement for long-term antiplatelet therapy. The initial two-year results had suggested a numerically higher rate of all-cause mortality in the clopidogrel group however, though the trial was not powered for mortality, that trend was not borne out in the initial longer-term follow-up, nor this very long-term follow-up and therefore was likely simply a chance finding.
The lower thrombotic events were predominantly driven by lower rates of non-fatal stroke and admission with ACS. Whilst these may not translate into mortality differences between groups, both non-fatal strokes and recurrent ACS carry important implications for use of health care resources and more importantly, impact on patients functional capacity and quality of life in the longer term. The lower bleeding rates with clopidogrel were again predominantly due to lower rates of BARC 3 bleeding, which has more important clinical implications than BARC 2 bleeding. Interestingly, in the post-24-month period aspirin was discontinued for both gastrointestinal discomfort or minor bleeding and self-discontinued more frequently than clopidogrel. Given the importance of compliance with antiplatelet therapy this raises the question with regard to clopidogrel being a more acceptable medication to patients in the longer term.
Using a per-protocol analysis, results were similar to the intention-to-treat analysis however, the effect estimates were larger for the primary and secondary composite endpoints with lower numbers needed to treat. Whilst the per-protocol analysis is subject to bias, given that antiplatelets were stopped/changed at the discretion of the treating physician, it is supportive of long-term clopidogrel in patients who tolerate same.
As with any analysis, there are a number of limitations which must be borne in mind, most importantly the trial is not powered to assess outcomes in subgroups, though there appeared to be a larger reduction in risk amongst patients with chronic kidney disease. The study was carried out in exclusively Korean centres therefore, the results may not be generalisable to non-East Asian populations. Clopidogrel non-responsiveness remains a concern due to the risk of stent thrombosis however, platelet reactivity testing is not routinely recommended. Furthermore, by nature of the trial design, patients who had an ischaemic or haemorrhagic event in the 6-18 months post PCI will not have been enrolled in this study. Therefore, this strategy, presently, should only be considered in patients who are event-free at discontinuation of DAPT.
Overall, this trial further supports the benefit of monotherapy with a P2Y12 inhibitor above aspirin in this population post PCI. To date, the optimal long-term antiplatelet strategy in all populations remains undetermined, and clinicians must carefully assess individual patients bleeding and ischaemic risks when prescribing and reviewing antiplatelet therapy.
References
- Koo BK, Kang J, Park KW, Rhee TM, Yang HM, Won KB, et al. Aspirin versus clopidogrel for chronic maintenance monotherapy after percutaneous coronary intervention (HOST-EXAM): an investigator-initiated, prospective, randomised, open-label, multicentre trial. Lancet. 2021 May 14. doi:10.1016/S0140-6736(21)01063-1 PubMed PMID: 34010616.
- Kang J, Park KW, Lee H, Hwang D, Yang HM, Rha SW, et al. Aspirin vs. Clopidogrel for Chronic Maintenance Monotherapy after Percutaneous Coronary Intervention: the HOST-EXAM Extended Study. Circulation. 2022 Oct 24. Located at: Hagerstown, MD. doi:10.1161/CIRCULATIONAHA.122.062770
- Baber U, Dangas G, Angiolillo DJ, Cohen DJ, Sharma SK, Nicolas J, et al. Ticagrelor alone vs. ticagrelor plus aspirin following percutaneous coronary intervention in patients with non-ST-segment elevation acute coronary syndromes: TWILIGHT-ACS. Eur Heart J. 2020 Oct 1;41(37):3533–45. doi:10.1093/eurheartj/ehaa670 PubMed PMID: 33085967.
- Watanabe H, Domei T, Morimoto T, Natsuaki M, Shiomi H, Toyota T, et al. Effect of 1-Month Dual Antiplatelet Therapy Followed by Clopidogrel vs 12-Month Dual Antiplatelet Therapy on Cardiovascular and Bleeding Events in Patients Receiving PCI: The STOPDAPT-2 Randomized Clinical Trial. JAMA. 2019 Jun 25;321(24):2414. doi:10.1001/jama.2019.8145
- Natsuaki M, Morimoto T, Yamamoto E, Shiomi H, Furukawa Y, Abe M, et al. One-year outcome of a prospective trial stopping dual antiplatelet therapy at 3 months after everolimus-eluting cobalt-chromium stent implantation: ShortT and OPtimal duration of Dual AntiPlatelet Therapy after everolimus-eluting cobalt-chromium stent (STOPDAPT) trial. Cardiovasc Interv and Ther. 2016 Jul;31(3):196–209. doi:10.1007/s12928-015-0366-9
- Kim BK, Hong SJ, Cho YH, Yun KH, Kim YH, Suh Y, et al. Effect of Ticagrelor Monotherapy vs Ticagrelor With Aspirin on Major Bleeding and Cardiovascular Events in Patients With Acute Coronary Syndrome: The TICO Randomized Clinical Trial. JAMA. 2020 Jun 16;323(23):2407–16. doi:10.1001/jama.2020.7580 PubMed PMID: 32543684; PubMed Central PMCID: PMC7298605.
- Hong SJ, Lee SJ, Suh Y, Yun KH, Kang TS, Shin S, et al. Stopping Aspirin Within 1 Month After Stenting for Ticagrelor Monotherapy in Acute Coronary Syndrome: The T-PASS Randomized Noninferiority Trial. Circulation. 2024 Feb 20;149(8):562–73. doi:10.1161/CIRCULATIONAHA.123.066943 PubMed PMID: 37878786.




No comments yet!