Six-year outcomes after transcatheter vs surgical aortic valve replacement in low-risk patients with aortic stenosis
Selected in JACC by A. Sticchi
The 6-year results of the Evolut Low Risk trial confirm the absence of significant differences in the composite primary endpoint between TAVR and surgery, but introduce an important nuance: the first signal of increased valve reintervention in the TAVR arm within this trial.
References
Authors
John K. Forrest, Steven J. Yakubov, G. Michael Deeb, and Michael J. Reardon
Reference
2026;10.1016/j.jacc.2026.02.5063
Published
16 February 2026
Link
Read the abstractReviewer
Latest contributions
Two-year outcomes after transcatheter tricuspid repair without cross-over in the randomised TRI-FR trial Six-year outcomes after transcatheter vs surgical aortic valve replacement in low-risk patients with aortic stenosis Protect the head to head trial: Randomized comparison of emboliner embolic protection catheter vs. sentinel cerebral protection system during trans...My Comment
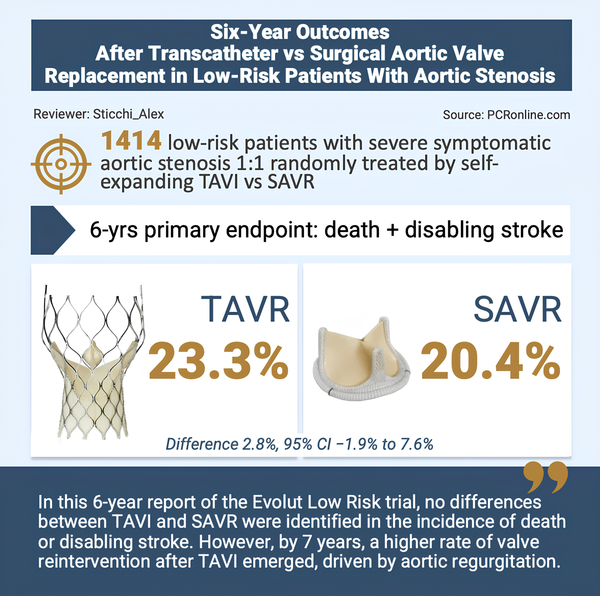
Visual summarising key information from the 6-year report of the Evolut Low Risk trial -
Courtesy of Alex Sticchi for PCRonline
Why this study – the rationale/objective?
TAVR has become the dominant treatment strategy for severe symptomatic aortic stenosis across the spectrum of surgical risk. The landmark Evolut Low Risk and PARTNER 3 trials demonstrated non-inferiority of TAVR compared with surgery in low-risk patients, leading to the FDA expansion of the TAVR indication and subsequent guideline endorsement on both sides of the Atlantic.
However, in a low-risk population with relatively longer life expectancy, long-term valve durability becomes a central concern. Previous reports from the Evolut Low Risk trial at 2, 3, 4, and 5 years demonstrated comparable outcomes between the two strategies, with no significant differences in the composite of all-cause mortality or disabling stroke, cardiovascular mortality, or reintervention rates1-4.
The 2025 ESC/EACTS guidelines further lowered the age cutoff for TAVR eligibility5, reinforcing the trend toward transcatheter treatment in younger patients.
In this context, the 6-year follow-up of the Evolut Low Risk trial was prespecified to assess ongoing safety and efficacy. Importantly, this analysis revealed for the first time an increased reintervention rate in the TAVR arm, prompting the investigators to accelerate reporting and conduct additional exploratory analyses on available 7-year data to characterise the incidence, aetiology, and clinical impact of this emerging signal.
How was it executed? The methodology
The Evolut Low Risk trial (NCT02701283) is an international, prospective, multicentre, randomised trial conducted at 87 sites across seven countries (Australia, Canada, France, Japan, the Netherlands, New Zealand, and the United States). Low-risk patients with severe tri-leaflet symptomatic aortic stenosis (predicted surgical mortality < 3 %) were randomised 1:1 to TAVR with a supra-annular self-expanding bioprosthesis (CoreValve, Evolut R, or Evolut PRO; Medtronic) or surgical aortic valve replacement. Procedures were performed between March 2016 and May 2019, with annual follow-up planned for 10 years.
A total of 1,414 patients underwent attempted implantation (730 TAVR, 684 surgery). The TAVR valve distribution was: Evolut R 72.9 %, Evolut PRO 23.5 %, and CoreValve 3.6 %. Notably, these devices are no longer commercially available in the United States, having been succeeded by Evolut PRO+, Evolut FX, and Evolut FX+.
The pre-specified 6-year analysis reported Kaplan-Meier estimates with log-rank test for the primary composite endpoint (all-cause mortality or disabling stroke) and key secondary endpoints. A vital status sweep was conducted to ascertain mortality in patients lost to follow-up or withdrawn. Given the emerging re-intervention signal at 6 years, the authors performed additional post hoc analyses on available 7-year data (555 TAVR and 480 surgery patients) using cumulative incidence functions to account for the competing risk of death. An independent Clinical Events Committee adjudicated endpoint-related adverse events. Echocardiographic assessments were adjudicated by a core laboratory at years 1–5, 7, and 10 per protocol; therefore, no core laboratory echocardiographic data were available at 6 years.
What is the main result?
At 6 years, the composite endpoint of all-cause mortality or disabling stroke was similar between groups: 23.3 % for TAVR and 20.4 % for surgery (difference: 2.8 %; 95 % CI: −1.9 % to 7.6 %; P = 0.43). Cardiovascular mortality was virtually identical (11.1 % vs 11.0 %; P = 0.81), while non-cardiovascular mortality showed a non-significant numerical trend favouring surgery (11.0 % vs 8.7 %; P = 0.24). All-cause mortality with vital status sweep was 23.3 % for TAVR and 20.2 % for surgery (P = 0.24). At 7 years, no significant differences in all-cause mortality (27.7 % vs 23.9 %; P = 0.29) or cardiovascular mortality (15.9 % vs 14.1 %; P = 0.73) were observed.
The pivotal finding was the emergence of a higher re-intervention rate after TAVR. The cumulative incidence of re-intervention at 6 years was 5.5 % for TAVR and 3.3 % for surgery (sHR: 1.66; 95 % CI: 0.96–2.86; P = 0.07), reaching statistical significance at 7 years (9.8 % vs 6.0 %; sHR: 1.68; 95 % CI: 1.10–2.58; P = 0.02). This difference was entirely driven by re-intervention due to aortic regurgitation (5.6 % vs 1.6 % at 7 years; sHR: 3.39; 95 % CI: 1.62–7.07; P < 0.001), whereas re-intervention for stenosis was comparable (3.6 % vs 3.5 %; P = 0.70).
An exploratory post-dilation analysis showed that off-guidance post-dilation (balloon exceeding valve waist per the 2020 IFU update) was associated with a 9.3 % rate of regurgitation-related re-intervention, compared with 4.7 % for all other TAVR (P = 0.02). The highest rate was observed in the Evolut 34R with off-guidance post-dilation (19.1 %). However, re-interventions occurred across all valve sizes and post-dilation categories, and rates remained numerically higher than surgery (1.6 %) even without postdilation (4.8 %).
Other notable 6-year results included: persistently higher permanent pacemaker implantation with TAVR (29.4 % vs 13.4 %; P < 0.001), lower atrial fibrillation with TAVR (18.1 % vs 42.3 %; P < 0.001), and lower valve endocarditis with TAVR (1.4 % vs 3.2 %; P = 0.04). Among the 60 patients requiring re-intervention after TAVR, 30-day mortality was 3.3 % (2/60), contrasting with the 13.1 % reported in the EXPLANT-TAVR registry6; 65 % of TAVR re-interventions required surgical explant.
Critical reading and the relevance for clinical practice:
The 6-year results of the Evolut Low Risk trial confirm the absence of significant differences in the composite primary endpoint between TAVR and surgery, but introduce an important nuance: the first signal of increased valve re-intervention in the TAVR arm within this trial. This finding warrants careful interpretation on multiple levels.
First, in absolute terms, the difference in re-intervention rates remains modest. At 7 years, more than 90 out of 100 TAVR patients and 94 out of 100 surgical patients did not require re-intervention. Nevertheless, in a low-risk population with a mean age of 74 years at implantation and substantial remaining life expectancy, even small differences may compound over time and carry meaningful clinical consequences, particularly if further divergence is observed during 8–10-year follow-up. The event curves crossing at 5 years for the composite primary endpoint, although not statistically significant, mirror a similar phenomenon observed in PARTNER 3 at year 3, where curves subsequently stabilised7. Whether a similar stabilisation will occur in this trial remains to be determined.
Second, the re-intervention phenotype matters as much as incidence. As underlined by Gupta and Cohen in the accompanying editorial9, from a patient’s perspective the type of re-intervention is critical: 65 % of TAVR re-interventions required surgical explant—the very procedure many patients believed they were going to avoid. Although 30-day mortality was low in this trial population (3.3 %), real-world explant mortality is considerably higher, as demonstrated by the EXPLANT-TAVR registry6. This asymmetry between expectations and reality is particularly important in the context of shared decision-making.
Furthermore, the clinical behaviour of the two re-intervention aetiologies differs: while stenosis typically evolves gradually and can be tracked via surveillance imaging, clinically significant transvalvular regurgitation may present more abruptly and with limited warning, adding further complexity to patient monitoring strategies.
Third, the mechanistic link between off-guidance post-dilation and regurgitation-related re-intervention is biologically plausible but remains circumstantial. Bench testing has demonstrated that balloon oversizing beyond the valve waist can injure prosthetic leaflets at their attachment margins8, and the 2020 Field Corrective Action was issued accordingly. However, as Gupta and Cohen emphasise9, post-dilation is not a random event: it is typically driven by suboptimal valve expansion or residual paravalvular leak, conditions that often reflect greater anatomic complexity such as asymmetric or heavy annular calcification. Off-guidance post-dilation may therefore function both as a cause of valve injury and as a marker for a challenging substrate. Without detailed insight into the baseline anatomy and intraprocedural valve performance of patients who required re-intervention, attributing premature valve failure to a single mechanism should remain appropriately cautious. Importantly, the majority of regurgitation-related re-interventions occurred in valves that were not subjected to off-guidance post-dilation, suggesting that platform-inherent characteristics play an independent role.
Fourth, these results must be contextualised alongside other long-term data. A central message from the editorial is that neither TAVR nor SAVR is a monolith: durability may be device-specific, and the relevant unit of assessment is increasingly defined by the combination of device, procedure, and failure phenotype9. The NOTION trial reported no difference in re-intervention or valve failure at 10 years10, and the PARTNER 3 trial at 7 years demonstrated comparable reintervention rates (6.7 % vs 6.0 %) and bioprosthetic valve failure (6.9 % vs 7.3 %) between TAVR with the Sapien 3 balloon-expandable valve and surgery7. These differing signals between trials raise important questions about device-specific differences in long-term performance. However, indirect cross-trial comparison requires caution, as heterogeneity may also derive from differences in surgical valve selection, patient populations, and follow-up methodologies. Notably, some surgical bioprostheses commonly used during the low-risk trials (e.g., Trifecta) have demonstrated premature failure11, meaning that “SAVR durability” in these trials represents an average across multiple prosthesis designs with distinct performance profiles.
Fifth, re-intervention is an imperfect surrogate for valve performance. As highlighted by the editorial9, mortality can mask failure in high-risk patients, while delayed thresholds for redo procedures can obscure dysfunction in low-risk patients. The field has progressively moved toward harmonised definitions of bioprosthetic valve dysfunction and failure, as codified by the VARC-3 consensus12. A late re-intervention signal such as described here should prompt deeper phenotyping to link haemodynamic trajectories and anatomic findings to clinical events, rather than relying solely on reintervention or death as endpoints. The absence of core-laboratory echocardiographic data at 6 years in this trial is therefore a meaningful limitation, as it prevents a comprehensive assessment of subclinical structural valve deterioration in both arms.
Finally, this report highlights the critical role of lifetime management in patients with aortic stenosis. For younger, low-risk patients, decisions should consider not only leaflet durability but also technical factors including commissural alignment, implant depth, and neoskirt height that impact the feasibility of future coronary and valvular interventions13. Meticulous pre-procedure CT planning and valve selection are critical in determining a patient’s lifetime trajectory after TAVR.
Several methodological limitations deserve acknowledgement. The 7-year data are incomplete (76 % TAVR, 70 % surgery), the re-intervention analyses are post hoc and exploratory, no echocardiographic data were available at 6 years per protocol design, and no adjustment for multiplicity was performed. The industrial sponsorship (Medtronic owned the database and performed statistical analyses) should also be considered, although the authors report full interpretive and publication autonomy.
In summary, these findings should not dissuade thoughtful use of TAVR in low-risk patients, but they reinforce the critical role of Heart Team-based discussions that transparently address both known benefits and remaining uncertainties. Moreover, this report exemplifies the importance of high-quality clinical trials and transparent reporting of clinically relevant outcomes in shaping the future of valvular interventions.
The Evolut Low Risk trial 10-year report will be decisive in determining whether the observed re-intervention signal translates into meaningful clinical consequences or represents a self-limited phenomenon related to early-generation devices and procedural practices that have since evolved.
References
- Popma JJ, Deeb GM, Yakubov SJ, et al. Transcatheter aortic-valve replacement with a self-expanding valve in low-risk patients. N Engl J Med. 2019;380:1706–1715.
- Forrest JK, Deeb GM, Yakubov SJ, et al. 2-year outcomes after transcatheter versus surgical aortic valve replacement in low-risk patients. J Am Coll Cardiol. 2022;79:882–896.
- Forrest JK, Deeb GM, Yakubov SJ, et al. 3-year outcomes after transcatheter or surgical aortic valve replacement in low-risk patients with aortic stenosis. J Am Coll Cardiol. 2023;81:1663–1674.
- Forrest JK, Yakubov SJ, Deeb GM, et al. 5-year outcomes after transcatheter or surgical aortic valve replacement in low-risk patients with aortic stenosis. J Am Coll Cardiol. 2025;85:1523–1532.
- Praz F, Borger MA, Lanz J, et al. 2025 ESC/EACTS Guidelines for the management of valvular heart disease. Eur Heart J. 2025;ehaf194.
- Zaid S, Hirji SA, Bapat VN, et al. Surgical explantation of failed transcatheter aortic valve replacement. Ann Thorac Surg. 2023;116:933–942.
- Leon MB, Mack MJ, Pibarot P, et al. Transcatheter or surgical aortic-valve replacement in low-risk patients at 7 years. N Engl J Med. 2025. DOI: 10.1056/NEJMoa2509766.
- de Backer OSR, Stublaski R, Tadros H, et al. Postdilation of Evolut transcatheter heart valves: insights from bench testing. JACC Cardiovasc Interv. 2026;19:517–519.
- Gupta A, Cohen DJ. When the curve bends: Low-risk TAVR beyond year five. JACC. 2026. DOI: 10.1016/j.jacc.2026.02.5066.
- Thyregod HGH, Jørgensen TH, Ihlemann N, et al. Transcatheter or surgical aortic valve implantation: 10-year outcomes of the NOTION trial. Eur Heart J. 2024;45:1116–1124.
- US FDA. Abbott Trifecta Valves: Potential Risk of Early Structural Valve Deterioration – Letter to Health Care Providers. 2023.
- Généreux P, Piazza N, Alu MC, et al. Valve Academic Research Consortium 3: Updated Endpoint Definitions for Aortic Valve Clinical Research. JACC. 2021;77(21):2717–2746.
- Tang GHL, Amat-Santos IJ, De Backer O, et al. Rationale, Definitions, Techniques, and Outcomes of Commissural Alignment in TAVR. JACC Cardiovasc Interv. 2022;15(15):1497–1518.





No comments yet!